Advertisements
Advertisements
प्रश्न
Give one use of electromagnetic radiation in Ultraviolet radiation.
उत्तर
Ultraviolet radiation is used for sterilizing purposes.
APPEARS IN
संबंधित प्रश्न
A plane electromagnetic wave travels in vacuum along z-direction. What can you say about the directions of its electric and magnetic field vectors? If the frequency of the wave is 30 MHz, what is its wavelength?
Name the high energetic invisible electromagnetic waves which help in the study of the structure of crystals
In a Coolidge tube, electrons strike the target and stop inside it. Does the target get more and more negatively charged as time passes?
For harder X-rays,
(a) the wavelength is higher
(b) the intensity is higher
(c) the frequency is higher
(d) the photon energy is higher.
The electric current in an X-ray tube (from the target to the filament) operating at 40 kV is 10 mA. Assume that on an average, 1% of the total kinetic energy of the electron hitting hte target are converted into X-rays.
(a) What is the total power emitted as X-rays and (b) how much heat is produced in the target every second?
The short-wavelength limit shifts by 26 pm when the operating voltage in an X-ray tube is increased to 1.5 times the original value. What was the original value of the operating voltage?
(Use Planck constant h = 6.63 × 10-34 Js= 4.14 × 10-15 eVs, speed of light c = 3 × 108 m/s.)
Answer briefly.
Why are microwaves used in radar?
An e.m. wave exerts pressure on the surface on which it is incident. Justify.
Following QN ∴ 14, the radiation force on the roof will be
In an atom X, electrons absorb the energy from an external source. This energy “excites” the electrons from a lower-energy level to a higher-energy level around the nucleus of the atom. When electrons return to the ground state, they emit photons.
The figure below is the energy level diagram of atom X with three energy levels, E1 = 0.00eV, E2 = 1.78eV and E3 = 2.95eV. The ground state is considered 0 eV for reference. The transition of electrons takes place between levels E1 and E2.
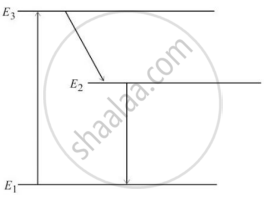
- What wavelength of radiation is needed to excite the atom to energy level E2 from E1?
- Suppose the external source has a power of 100 W. What would be the rate of photon emission?
