Advertisements
Advertisements
प्रश्न
Give plausible explanation for the following:
Cyclohexanone forms cyanohydrin in good yield but 2, 2, 6 trimethylcyclohexanone does not.
उत्तर
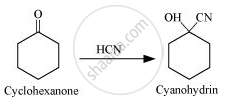
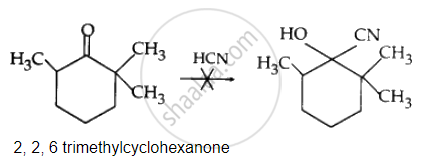
Due to the presence of three methyl groups at α-places, nucleophilic attack of CN– ions does not occur. This steric hindrance is absent in cyclohexane. Hence, nucleophilic attack of CN– ions occurs quickly. Hence, cyclohexanone cyanohydrin is obtained in good yield.
APPEARS IN
संबंधित प्रश्न
Arrange the following compound in increasing order of its reactivity in nucleophilic addition reactions.
Ethanal, Propanal, Propanone, Butanone.
Hint: Consider steric effect and electronic effect.
What is meant by the following term? Give an example of the reaction in the following case.
Imine
Acetaldehyde, Acetone, Di-tert-butyl ketone, Methyl tert-butyl ketone (reactivity towards HCN)
Complete the synthesis by giving missing starting material, reagent or product.
\[\ce{C6H5CHO ->[H2NCONHNH2]}\]
Write balanced chemical equations for action of ammonia on - acetone
Write the main product formed when propanal reacts with the following reagents:
2 moles of 3 CH OH in presence of dry HCl
How will you convert benzoic acid to m-bromobenzoic acid?
Benzaldehyde can be obtained from benzal chloride. Write reactions for obtaining benzalchloride and then benzaldehyde from it.
Carboxylic acids contain carbonyl group but do not show the nucleophilic addition reaction like aldehydes or ketones. Why?
Grignard reagent on reaction with acetone forms.
Write the name of product formed, when acetone is treated with 2, 4-dinitrophenyl hydrazine.
The increasing order of the following compounds towards HCN addition is:
| (i) | 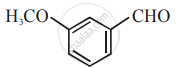 |
| (ii) | 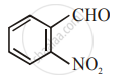 |
| (iii) | 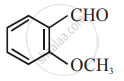 |
| (iv) | 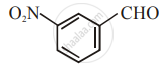 |
Aldehydes and ketones react with hydroxylamine to form ______.
Write the structure of the product formed when acetone reacts with 2, 4 DNP reagent.
Draw structure of the following derivative.
Acetaldehydedimethylacetal
Draw the structure of the following derivative.
The ethylene ketal of hexan-3-one
Draw structure of the following derivative:
Acetaldehydedimethylacetal
Give an example of the reaction in the following case.
Imine
