Advertisements
Advertisements
प्रश्न
Carboxylic acids contain carbonyl group but do not show the nucleophilic addition reaction like aldehydes or ketones. Why?
उत्तर
It is due to resonance as shown below. The partial positive charge on the carbonyl carbon atom is reduced
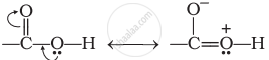
Similarly, a carbonyl group of aldehydes and ketones may be regarded as the resonance hybrid of the following structures.
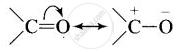
The carbonyl carbon of carboxylic group is less electrophilic than carbonyl carbon in aldehydes and ketones and hence nucleophilic addition reactions of aldehydes and ketones do not takes place with carboxylic acids.
APPEARS IN
संबंधित प्रश्न
Write the products formed when CH3CHO reacts with the following reagents : H2N – OH
Arrange the following compound in increasing order of its reactivity in nucleophilic addition reactions.
Benzaldehyde, p-Tolualdehyde, p-Nitrobenzaldehyde, Acetophenone.
Hint: Consider steric effect and electronic effect.
Write the structure of Phenylmethanamine.
Which of the following compounds is most reactive towards nucleophilic addition reactions?
Alkenes 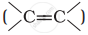 and carbonyl compounds
and carbonyl compounds 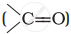 , both contain a π bond but alkenes show electrophilic addition reactions whereas carbonyl compounds show nucleophilic addition reactions. Explain.
, both contain a π bond but alkenes show electrophilic addition reactions whereas carbonyl compounds show nucleophilic addition reactions. Explain.
What happens when ethanal is treated with excess ethanol and acid?
Which of the following is most reactive in nucleophilic addition reactions?
Write the structure of the product formed when acetone reacts with 2, 4 DNP reagent.
Draw structures of the given derivatives.
The ethylene ketal of hexan-3-one
Draw structure of the following derivative:
Acetaldehydedimethylacetal
