Advertisements
Advertisements
प्रश्न
Carboxylic acids contain carbonyl group but do not show the nucleophilic addition reaction like aldehydes or ketones. Why?
उत्तर
It is due to resonance as shown below. The partial positive charge on the carbonyl carbon atom is reduced
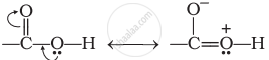
Similarly, a carbonyl group of aldehydes and ketones may be regarded as the resonance hybrid of the following structures.
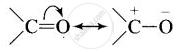
The carbonyl carbon of carboxylic group is less electrophilic than carbonyl carbon in aldehydes and ketones and hence nucleophilic addition reactions of aldehydes and ketones do not takes place with carboxylic acids.
APPEARS IN
संबंधित प्रश्न
Arrange the following compound in increasing order of its reactivity in nucleophilic addition reactions.
Ethanal, Propanal, Propanone, Butanone.
Hint: Consider steric effect and electronic effect.
Predict the product of the following reaction:
\[\begin{array}{cc}
\phantom{..............}\ce{O}\\
\phantom{..............}||\\
\ce{R - CH = CH - CHO + NH2 - C - NH - NH2 ->[H+]}\end{array}\]
What is meant by the following term? Give an example of the reaction in the following case.
Ketal
Predict the products formed when cyclohexanecarbaldehyde reacts with the following reagents.
Excess ethanol and acid
How are the following compounds prepared?
benzaldehyde from benzoyl chloride
Write balanced chemical equations for action of ammonia on - acetone
Out of p-tolualdehyde and p-nitrobenzaldehyde, which one is more reactive towards nucleophilic addition reactions, why?
Draw structures of the following derivative.
The ethylene ketal of hexan-3-one
Draw the structure of the following derivative.
Acetaldehydedimethylacetal
Give an example of the reaction in the following case.
Imine
