Advertisements
Advertisements
प्रश्न
Out of p-tolualdehyde and p-nitrobenzaldehyde, which one is more reactive towards nucleophilic addition reactions, why?
उत्तर
P-nitrobenzaldehyde is more reactive towards the nucleophilic addition reaction than p- tolualdehyde as Nitro group is electron withdrawing in nature. Presence of nitro group decrease electron density, hence facilitates the attack of nucleophile. The presence of \[\ce{-CH3}\] leads to +I effect as \[\ce{-CH3}\] is electron releasing group.
APPEARS IN
संबंधित प्रश्न
Write the products formed when CH3CHO reacts with the following reagents : H2N – OH
Predict the products formed when cyclohexanecarbaldehyde reacts with the following reagents.
Excess ethanol and acid
Complete the synthesis by giving missing starting material, reagent or product.

Write balanced chemical equations for action of ammonia on - acetone
Give a simple chemical test to distinguish between 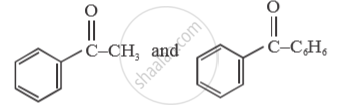
Write the main product formed when propanal reacts with the following reagents:
2 moles of 3 CH OH in presence of dry HCl
Write the main product formed when propanal reacts with the following reagents:
H2N- NH2 followed by heating with KOH in ethylene glycol.
How will you convert benzoic acid to m-bromobenzoic acid?
Draw structure of the following derivative.
The ethylene ketal of hexan-3-one
Draw structure of the following derivative.
The ethylene ketal of hexan-3-one
