Advertisements
Advertisements
Question
Out of p-tolualdehyde and p-nitrobenzaldehyde, which one is more reactive towards nucleophilic addition reactions, why?
Solution
P-nitrobenzaldehyde is more reactive towards the nucleophilic addition reaction than p- tolualdehyde as Nitro group is electron withdrawing in nature. Presence of nitro group decrease electron density, hence facilitates the attack of nucleophile. The presence of \[\ce{-CH3}\] leads to +I effect as \[\ce{-CH3}\] is electron releasing group.
APPEARS IN
RELATED QUESTIONS
Predict the products of the following reactions:
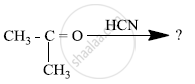
What is meant by the following term? Give an example of the reaction in the following case.
Oxime
Predict the products formed when cyclohexanecarbaldehyde reacts with the following reagents.
Excess ethanol and acid
How are the following compounds prepared?
benzaldehyde from benzoyl chloride
Acetone, Acetaldehyde, Benzaldehyde, Acetophenone – reactivity towards addition of HCN.
The pH of blood does not appreciably change by a small addition of acid or base because
Acetaldehyde and acetone differ in their reaction with
Draw structures of the given derivatives.
The ethylene ketal of hexan-3-one
Draw structure of the following derivative.
The ethylene ketal of hexane-3-one
Draw structure of the following derivative.
The ethylene ketal of hexan-3-one
