Advertisements
Advertisements
प्रश्न
Arrange the following compound in increasing order of its reactivity in nucleophilic addition reactions.
Ethanal, Propanal, Propanone, Butanone.
Hint: Consider steric effect and electronic effect.
उत्तर
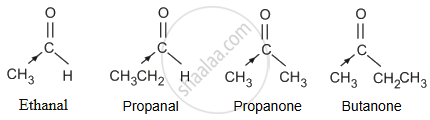
The +I effect of the alkyl group increases in the order:
Ethanal < Propanal < Propanone < Butanone
The electron density at the carbonyl carbon increases with the increase in the +I effect. As a result, the chances of attack by a nucleophile decrease. Hence, the increasing order of the reactivities of the given carbonyl compounds in nucleophilic addition reactions is:
Butanone < Propanone < Propanal < Ethanal
APPEARS IN
संबंधित प्रश्न
Write the structures of the main products when acetone (CH3 − CO − CH3) reacts with the following reagent :
H2N − NHCONH2/H+
Write the products formed when CH3CHO reacts with the following reagents : H2N – OH
Predict the product of the following reaction:
What is meant by the following term? Give an example of the reaction in the following case.
Schiff’s base
Predict the products formed when cyclohexanecarbaldehyde reacts with the following reagents.
Semicarbazide and weak acid
Acetaldehyde, Acetone, Di-tert-butyl ketone, Methyl tert-butyl ketone (reactivity towards HCN)
How will you bring about the following conversion in not more than two steps?
Bromobenzene to 1-Phenylethanol
Give plausible explanation for the following:
There are two −NH2 groups in semicarbazide. However, only one is involved in the formation of semicarbazones.
Give a simple chemical test to distinguish between 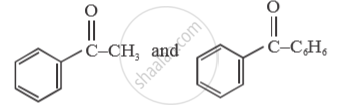
Identify the compounds A, B and C in the following reaction.
\[\ce{CH3 - Br ->[Mg/ether] (A) ->[(i) CO][(ii) Water] (B) ->[CH3OH/H+][Δ] (C)}\]
Write the name of product formed, when acetone is treated with 2, 4-dinitrophenyl hydrazine.
Aldehydes and ketones react with hydroxylamine to form ______.
Draw structures of the given derivatives.
The ethylene ketal of hexan-3-one
Draw structures of the given derivatives.
The ethylene ketal of hexan-3-one
Draw structure of the following derivative.
The ethylene ketal of hexane-3-one
Why dissociation of HCN is suppressed by the addition of HCL?
The product of the following reaction is
\[\begin{array}{cc}
\ce{O}\phantom{.........}\\
||\phantom{.........}\\
\ce{C2H5 - C - CH3 ->[H2/Ni][\Delta] \phantom{..}?}\end{array}\]
Draw the structure of the following derivative.
The ethylene ketal of hexan-3-one
Draw the structure of the given derivative.
The ethylene ketal of hexan-3-one
