Advertisements
Advertisements
प्रश्न
Alkenes 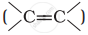 and carbonyl compounds
and carbonyl compounds 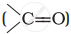 , both contain a π bond but alkenes show electrophilic addition reactions whereas carbonyl compounds show nucleophilic addition reactions. Explain.
, both contain a π bond but alkenes show electrophilic addition reactions whereas carbonyl compounds show nucleophilic addition reactions. Explain.
उत्तर
Carbonyl group is polar in nature. Due to larger electronegativity of oxygen as compared to carbon, carbon acquires partial positive charge while O acquires partial negative charge.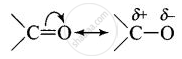
Because of slight positive charge on C atom, it is attacked by nucleophiles and, therefore, undergoes nucleophilic addition reaction.
Ethylenic double bond is a non-polar bond and is a source of electrons. Therefore, it is attacked by electrophiles and undergoes electrophilic addition reactions.
APPEARS IN
संबंधित प्रश्न
Arrange the following compound in increasing order of its reactivity in nucleophilic addition reactions.
Benzaldehyde, p-Tolualdehyde, p-Nitrobenzaldehyde, Acetophenone.
Hint: Consider steric effect and electronic effect.
What is meant by the following term? Give an example of the reaction in the following case.
Cyanohydrin
What is meant by the following term? Give an example of the reaction in the following case.
Oxime
Complete the synthesis by giving missing starting material, reagent or product.

Write balanced chemical equations for action of ammonia on - acetone
Which of the following compounds is most reactive towards nucleophilic addition reactions?
Which of the following has the most acidic hydrogen?
Paraldehyde is formed as a result of polymerisation:-
In the following reaction
\[\ce{Carbonyl compound + MeOH <=>[HCl] acetal}\]
Rate of the reaction is the highest for ______.
Draw structure of the following derivative:
Acetaldehydedimethylacetal
