Advertisements
Advertisements
प्रश्न
Which of the following is most reactive in nucleophilic addition reactions?
विकल्प
HCHO
CH3CHO
CH3COCH3
CH3COC2H5
उत्तर
HCHO
Explanation:
The group linked to carbonyl carbon influences reactivity in nucleophilic addition processes. The more electron-deficient the carbon, the greater its reactivity. The presence of alkyl groups reduces reactivity by lowering electron deficit. As a result, HCHO is more reactive towards nucleophilic addition processes.
APPEARS IN
संबंधित प्रश्न
Predict the product of the following reaction:
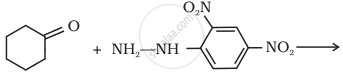
Write balanced chemical equations for action of ammonia on - acetone
Which of the following is the correct representation for intermediate of nucleophilic addition reaction to the given carbonyl compound (A):

(i) 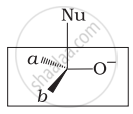
(ii) 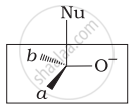
(iii) 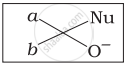
(iv) 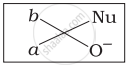
Alkenes 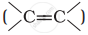 and carbonyl compounds
and carbonyl compounds 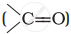 , both contain a π bond but alkenes show electrophilic addition reactions whereas carbonyl compounds show nucleophilic addition reactions. Explain.
, both contain a π bond but alkenes show electrophilic addition reactions whereas carbonyl compounds show nucleophilic addition reactions. Explain.
Identify the compounds A, B and C in the following reaction.
\[\ce{CH3 - Br ->[Mg/ether] (A) ->[(i) CO][(ii) Water] (B) ->[CH3OH/H+][Δ] (C)}\]
Acetaldehyde and acetone differ in their reaction with
Paraldehyde is formed as a result of polymerisation:-
Write the name of product formed, when acetone is treated with 2, 4-dinitrophenyl hydrazine.
The increasing order of the following compounds towards HCN addition is:
| (i) | 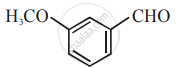 |
| (ii) | 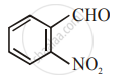 |
| (iii) | 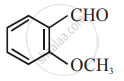 |
| (iv) | 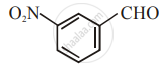 |
The product of the following reaction is
\[\begin{array}{cc}
\ce{O}\phantom{.........}\\
||\phantom{.........}\\
\ce{C2H5 - C - CH3 ->[H2/Ni][\Delta] \phantom{..}?}\end{array}\]
