Advertisements
Advertisements
प्रश्न
Which of the following reactions are feasible?
विकल्प
\[\ce{CH3CH2Br + Na^{+}O^{-}C(CH3)3 -> CH3CH2 - O - C(CH3)3}\]
\[\ce{(CH3)3 C - Cl + Na^{+}O^{-}CH2CH3 -> CH3CH2 - O - C(CH3)3}\]
Both \[\ce{CH3CH2Br + Na^{+}O^{-}C(CH3)3 -> CH3CH2 - O - C(CH3)3}\] and \[\ce{(CH3)3 C - Cl + Na^{+}O^{-}CH2CH3 -> CH3CH2 - O - C(CH3)3}\]
Neither \[\ce{CH3CH2Br + Na^{+}O^{-}C(CH3)3 -> CH3CH2 - O - C(CH3)3}\] nor \[\ce{(CH3)3 C - Cl + Na^{+}O^{-}CH2CH3 -> CH3CH2 - O - C(CH3)3}\]
उत्तर
\[\ce{CH3CH2Br + Na^{+}O^{-}C(CH3)3 -> CH3CH2 - O - C(CH3)3}\]
Explanation:
The above reaction is an example of Williamson synthesis. In this process, an alkyl halide is allowed to react with sodium alkoxide. The reaction involves the SN2 attack of an alkoxide ion on a primary alkyl halide.
APPEARS IN
संबंधित प्रश्न
Explain the mechanism of the following reaction:
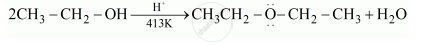
Account for the following :
t-butyl chloride on heating with sodium methoxide gives 2-methylpropene instead of t-butyl methyl ether.
Which of the following cannot be made by using Williamson Synthesis:
Preparation of alcohols from alkenes involves the electrophilic attack on alkene carbon atom. Explain its mechanism.
The Williamson ether synthesis produces ethers by reacting an
Write the mechanism of the following reaction:
\[\ce{2CH3CH2OH ->[H^+][413 K] CH3-CH2-O-CH2-CH3 + H2O}\]
What is metamerism? Give the structure and IUPAC name of metamers of 2-methoxy propane.
Write the name of reagent and equation for the preparation of the following ethers by Williamson’s synthesis:
2-Methoxy-2-methylpropane
Identify the product(s) is/are formed when 1-methoxy propane is heated with excess HI. Name the mechanism involved in the reaction.
Write the names of reagents and equations for the preparation of the following ether by Williamson’s synthesis:
2-Methoxy-2-methylpropane
