Advertisements
Advertisements
प्रश्न
Which of the following is the correct representation for intermediate of nucleophilic addition reaction to the given carbonyl compound (A):

(i) 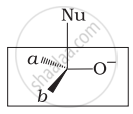
(ii) 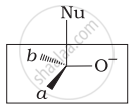
(iii) 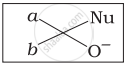
(iv) 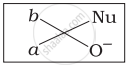
उत्तर
(i) 
(ii) 
Explanation:
The given compound is a planar molecule with sp2 hybridized carbon. Carbon atoms are attacked by nucleophiles. If the nucleophile approaches from the front, the (i) and (ii) molecules shift to the front and back positions, respectively and the carbon becomes tetrahedral. Another alternative is that (i) and (ii) molecules will be located above and below the plane, respectively. As a result, the choices (iii) and (iv) aren't represented as planar molecules.
APPEARS IN
संबंधित प्रश्न
How are the following compounds prepared?
benzaldehyde from benzene
Write the structures of the main products when acetone (CH3 − CO − CH3) reacts with the following reagent :
H2N − NHCONH2/H+
Arrange the following compound in increasing order of its reactivity in nucleophilic addition reactions.
Ethanal, Propanal, Propanone, Butanone.
Hint: Consider steric effect and electronic effect.
Write the structure of Phenylmethanamine.
Give a simple chemical test to distinguish between 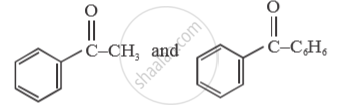
Write the main product formed when propanal reacts with the following reagents:
H2N- NH2 followed by heating with KOH in ethylene glycol.
A Idol condensation will not be observed in
What happens when ethanal is treated with excess ethanol and acid?
Draw the structure of the following derivative.
The ethylene ketal of hexan-3-one
Draw the structure of the following derivative.
Acetaldehydedimethylacetal
