Advertisements
Advertisements
प्रश्न
Why is there a large difference in the boiling points of butanal and butan-1-ol?
उत्तर
Butan-1 -ol has higher boiling point (391 K) than butanal due to intermolecular hydrogen bonding between –OH groups of alcohols.
APPEARS IN
संबंधित प्रश्न
How will you bring about the following conversion?
Ethanal to but-2-enal
How will you convert ethanal into the following compound?
But-2-enal
Write structural formulas and names of four possible aldol condensation products from propanal and butanal. In each case, indicate which aldehyde acts as nucleophile and which as electrophile.
How will you bring about the following conversion in not more than two steps?
Ethanol to 3-Hydroxybutanal
Why is alpha (α) hydrogen of carbonyl compounds acidic in nature?
Write chemical equations of the following reaction :
Benzoyl chloride is hydrogenated in the presence of `"Pd"/(BaSO_4)`
Cannizaro’s reaction is not given by ______.
Which product is formed when the compound 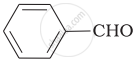 is treated with concentrated aqueous \[\ce{KOH}\] solution?
is treated with concentrated aqueous \[\ce{KOH}\] solution?
Compound ‘A’ was prepared by oxidation of compound ‘B’ with alkaline \[\ce{KMnO4}\]. Compound ‘A’ on reduction with lithium aluminium hydride gets converted back to compound ‘B’. When compound ‘A’ is heated with compound B in the presence of \[\ce{H2SO4}\] it produces fruity smell of compound C to which family the compounds ‘A’, ‘B’ and ‘C’ belong to?
Identify A and B from the following reaction:
\[\begin{array}{cc}
\ce{CH3}\phantom{.................}\\
|\phantom{....................}\\
\phantom{}\ce{2CH3 - C = O ->[Ba(OH)2] A ->[Δ] B + H2O}
\end{array}\]
