Advertisements
Advertisements
प्रश्न
How will you bring about the following conversion in not more than two steps?
Ethanol to 3-Hydroxybutanal
How will you convert the following:
Ethanol to 3-Hydroxybutanal
उत्तर १
\[\begin{array}{cc}
\phantom{...............................................................}\ce{OH}\\
\phantom{.............................................................}|\\
\ce{\underset{Ethanal}{CH3 - CH2 - OH}->[Mild oxidation][KMnO4/dil.H2SO4]\underset{(Two molecules)}{\underset{Acetaldehyde}{CH3 - CHO}}->[dil.OH-][warm]\underset{3-Hydroxybutanal}{CHO - CH2 - CH - CH3}}\
\end{array}\]
उत्तर २
\[\begin{array}{cc}
\phantom{....................................}\ce{OH}\\
\phantom{..................................}|\\
\ce{CH3CH2OH ->[PCC] CH3CHO ->[dil NaOH][Aldol condensation] \underset{3-Hydroxybutanal}{CH3 - CH - CH2 - CHO}}
\end{array}\]
PCC = Pyridinechlorochromate
APPEARS IN
संबंधित प्रश्न
What is meant by the following term? Give an example of the reaction in the following case.
Aldol
How will you convert ethanal into the following compound?
Butane-1, 3-diol
Describe the following:
Cross aldol condensation
Write chemical equations of the following reaction :
Benzoyl chloride is hydrogenated in the presence of `"Pd"/(BaSO_4)`
Which product is formed when the compound 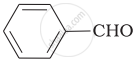 is treated with concentrated aqueous \[\ce{KOH}\] solution?
is treated with concentrated aqueous \[\ce{KOH}\] solution?
Which of the following compounds do not undergo aldol condensation?
(i) \[\ce{CH3 - CHO}\]
(ii) 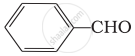
(iii) \[\begin{array}{cc}
\phantom{}\ce{O}\\
\phantom{}||\\
\ce{CH3 - C - CH3}
\end{array}\]
(iv) \[\begin{array}{cc}
\phantom{}\ce{CH3}\\
|\phantom{...}\\
\ce{CH3 - C - CHO}\phantom{..}\\
|\phantom{...}\\
\phantom{}\ce{CH3}\\
\end{array}\]
Why is there a large difference in the boiling points of butanal and butan-1-ol?
Why are carboxylic acids more acidic than alcohols or phenols although all of them have hydrogen atom attached to a oxygen atom \[\ce{(-O-H)}\]?
Assertion: The α-hydrogen atom in carbonyl compounds is less acidic.
Reason: The anion formed after the loss of α-hydrogen atom is resonance stabilised.
Give reasons to support the answer:
Presence of Alpha hydrogen in aldehydes and ketones is essential for aldol condensation.
Convert the following:
Acetaldehyde to But-2-enal
Predict the reagent for carrying out the following transformations:
Ethanal to 3-hydroxy butanal
The major product of the following reaction is:
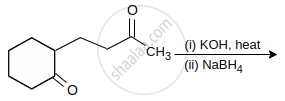
Write a note on the aldol condensation reaction of acetaldehyde.
What is aldol condensation? Explain it with suitable examples.
