Advertisements
Advertisements
प्रश्न
Assertion: The α-hydrogen atom in carbonyl compounds is less acidic.
Reason: The anion formed after the loss of α-hydrogen atom is resonance stabilised.
विकल्प
Assertion and reason both are correct and reason is correct explanation of assertion.
Assertion and reason both are wrong statements.
Assertion is correct statement but reason is wrong statement.
Assertion is wrong statement but reason is correct statement.
Assertion and reason both are correct statements but reasson is not correct explanation of assertion.
उत्तर
Assertion is wrong statement but reason is correct statement.
Explanation:
Because of the presence of electron-withdrawing carbonyl group, the alpha hydrogen atom in carbonyl compounds is acidic. In nature, hydrogen is very acidic.
Because the cation is released in the form of \[\ce{H-}\], the anion created after the loss of the -hydrogen atom is resonance stabilised.
APPEARS IN
संबंधित प्रश्न
Give reasons Acetylation of aniline reduces its activation effect.
Why is alpha (α) hydrogen of carbonyl compounds acidic in nature?
Write a chemical equation for the following reaction:
Propanone is treated with dilute Ba( OH)2.
What is substituted imine called?
Which product is formed when the compound 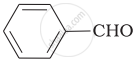 is treated with concentrated aqueous \[\ce{KOH}\] solution?
is treated with concentrated aqueous \[\ce{KOH}\] solution?
Cross aldol condensation occurs between
Predict the reagent for carrying out the following transformations:
Ethanal to 3-hydroxy butanal
Which of the following compounds will undergo self-condensation in the presence of dilute NaOH solution?
Why is the α-hydrogens of aldehydes and ketones are acidic in nature?

Identify A and B:
