Advertisements
Advertisements
प्रश्न
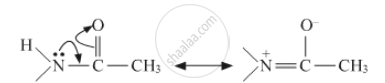
Give reasons Acetylation of aniline reduces its activation effect.
उत्तर
The lone pair of nitrogen will get involved in resonance with the carbonyl group. Hence it will reduce the activity of benzene ring in aniline. The resonance involved is shown below:
APPEARS IN
संबंधित प्रश्न
How will you bring about the following conversion?
Ethanal to but-2-enal
How will you convert ethanal into the following compound?
But-2-enal
Write structural formulas and names of four possible aldol condensation products from propanal and butanal. In each case, indicate which aldehyde acts as nucleophile and which as electrophile.
How will you bring about the following conversion in not more than two steps?
Benzaldehyde to 3-Phenylpropan-1-ol
Which of the following compounds do not undergo aldol condensation?
(i) \[\ce{CH3 - CHO}\]
(ii) 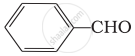
(iii) \[\begin{array}{cc}
\phantom{}\ce{O}\\
\phantom{}||\\
\ce{CH3 - C - CH3}
\end{array}\]
(iv) \[\begin{array}{cc}
\phantom{}\ce{CH3}\\
|\phantom{...}\\
\ce{CH3 - C - CHO}\phantom{..}\\
|\phantom{...}\\
\phantom{}\ce{CH3}\\
\end{array}\]
Why is there a large difference in the boiling points of butanal and butan-1-ol?
Explain Aldol condensation of ethanal.
Which of the following does not give aldol condensation reaction?

Identify A and B:
Write a note on the aldol condensation reaction of acetaldehyde.
