Advertisements
Advertisements
प्रश्न
How will you convert ethanal into the following compound?
But-2-enal
उत्तर १
\[\ce{\underset{Ethanal}{2CH3CHO}->[(i) dil.NaOH][(ii) H+, \Delta]\underset{But-2-enal}{CH3CH = CHCHO}}\]
उत्तर २
\[\ce{\underset{Ethanal}{2CH3CHO}->[dil.NaOH][Aldol condensation]\underset{Hydroxybutanal}{\overset{4}{C}H3 - \overset{3}{C}HOH - \overset{2}{C}H2 - \overset{1}{C}HO}->[H3O+/\Delta][-H2O]\underset{But-2-enal}{CH3 - CH = CH - CHO}}\]
APPEARS IN
संबंधित प्रश्न
Which of the following compounds would undergo aldol condensation, which the Cannizzaro reaction and which neither? Write the structures of the expected products of aldol condensation and Cannizzaro reaction.
- Methanal
- 2-Methylpentanal
- Benzaldehyde
- Benzophenone
- Cyclohexanone
- 1-Phenylpropanone
- Phenylacetaldehyde
- Butan-1-ol
- 2, 2-Dimethylbutanal
Write structural formulas and names of four possible aldol condensation products from propanal and butanal. In each case, indicate which aldehyde acts as nucleophile and which as electrophile.
How will you bring about the following conversion in not more than two steps?
Benzaldehyde to 3-Phenylpropan-1-ol
Complete the synthesis by giving missing starting material, reagent or product.
\[\begin{array}{cc}
\ce{C6H5CHO}\phantom{............}\\
\phantom{........}\ce{+\phantom{......}\ce{->[dil.NaOH][\Delta]}}\phantom{...}\\
\ce{CH3CH2CHO}\phantom{............}
\end{array}\]
Why is alpha (α) hydrogen of carbonyl compounds acidic in nature?
Write a chemical equation for the following reaction:
Propanone is treated with dilute Ba( OH)2.
Which product is formed when the compound 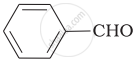 is treated with concentrated aqueous \[\ce{KOH}\] solution?
is treated with concentrated aqueous \[\ce{KOH}\] solution?
What product will be formed on reaction of propanal with 2-methylpropanal in the presence of \[\ce{NaOH}\]? What products will be formed? Write the name of the reaction also.
Assertion: The α-hydrogen atom in carbonyl compounds is less acidic.
Reason: The anion formed after the loss of α-hydrogen atom is resonance stabilised.
Assertion: Aromatic aldehydes and formaldehyde undergo Cannizaro reaction.
Reason: Aromatic aldehydes are almost as reactive as formaldehyde.
Give reasons to support the answer:
Presence of Alpha hydrogen in aldehydes and ketones is essential for aldol condensation.
Cross aldol condensation occurs between
Convert the following:
Acetaldehyde to But-2-enal
Explain Aldol condensation of ethanal.
Which of the following compounds will undergo self-condensation in the presence of dilute NaOH solution?
Write a note on the aldol condensation reaction of acetaldehyde.
