Advertisements
Advertisements
प्रश्न
Convert the following:
Acetaldehyde to But-2-enal
उत्तर
Aldol condensation (in presence of dil. \[\ce{NaOH}\]) followed by dehydration.
\[\begin{array}{cc}
\phantom{}\ce{\underset{Acetaldehyde}{CH3CHO} ->[dil. NaOH] CH3 - CH - CH2 - CHO ->[Δ][-H2O] \underset{\underset{(Adol condensation product)}{But-2-enal}}{CH3 - CH} = CH - CHO}\\
|\phantom{.........................}\\
\ce{\underset{\underset{(Adol)}{3-Hydroxybutanal}}{OH}}\phantom{.......................}\\
\end{array}\]
APPEARS IN
संबंधित प्रश्न
A compound 'A' of molecular formula C2H3OCl undergoes a series of reactions as shown below. Write the structures of A, B, C and D in the following reactions :
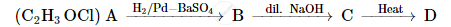
Write the products formed when CH3CHO reacts with the following reagents: CH3CHO in the presence of dilute NaOH
How will you convert ethanal into the following compound?
Butane-1, 3-diol
How will you bring about the following conversion in not more than two steps?
Ethanol to 3-Hydroxybutanal
Complete the synthesis by giving missing starting material, reagent or product.
\[\begin{array}{cc}
\ce{C6H5CHO}\phantom{............}\\
\phantom{........}\ce{+\phantom{......}\ce{->[dil.NaOH][\Delta]}}\phantom{...}\\
\ce{CH3CH2CHO}\phantom{............}
\end{array}\]
Compounds A and C in the following reaction are:
\[\ce{CH3CHO ->[(i) CH3MgBr][(ii) H2O] (A) ->[H2SO4, Δ] (B) ->[Hydroboration oxidation] (C)}\]
Cross aldol condensation occurs between
Identify A and B from the following reaction:
\[\begin{array}{cc}
\ce{CH3}\phantom{.................}\\
|\phantom{....................}\\
\phantom{}\ce{2CH3 - C = O ->[Ba(OH)2] A ->[Δ] B + H2O}
\end{array}\]
\[\ce{CH3-CH2-CHO ->[dil][alkali] Product}\]
The product in the above reaction is:

Identify A and B:
