Advertisements
Advertisements
प्रश्न
Compounds A and C in the following reaction are:
\[\ce{CH3CHO ->[(i) CH3MgBr][(ii) H2O] (A) ->[H2SO4, Δ] (B) ->[Hydroboration oxidation] (C)}\]
विकल्प
identical
positional isomers
functional isomers
optical isomers
उत्तर
positional isomers
Explanation:
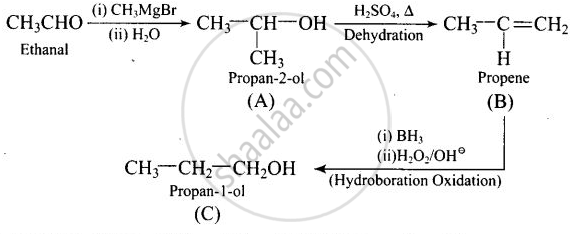
Thus, \[\begin{array}{cc}
\ce{CH3CH - OH}\\
|\phantom{....}\\
\ce{CH3}\phantom{..}
\end{array}\] and \[\ce{CH3 - CH2 - CH2OH}\] are positional isomers.
APPEARS IN
संबंधित प्रश्न
How will you convert ethanal into the following compound?
Butane-1, 3-diol
How will you convert ethanal into the following compound?
But-2-enal
Write structural formulas and names of four possible aldol condensation products from propanal and butanal. In each case, indicate which aldehyde acts as nucleophile and which as electrophile.
Write chemical equations of the following reaction :
Propanone is treated with dilute Ba (OH)2-.
Explain aldol condensation reaction in detail.
Cross aldol condensation occurs between
Which of the following does not give aldol condensation reaction?
Which of the following compounds will undergo self-condensation in the presence of dilute NaOH solution?
Why is the α-hydrogens of aldehydes and ketones are acidic in nature?
What is aldol condensation? Explain it with suitable examples.
