Advertisements
Advertisements
प्रश्न
Explain aldol condensation reaction in detail.
उत्तर
- Aldehydes containing at least one α–hydrogen atom undergo a reaction in presence of dilute alkali (dilute NaOH, KOH, or Na2CO3) as the catalyst to form β-hydroxy aldehydes (aldol). This reaction is known as the aldol reaction.
- Formation of aldol is an addition reaction.
- Aldol formed from aldehyde having α-hydrogens undergoes subsequent elimination of water molecule on warming. This gives rise to α,β-unsaturated aldehyde.
- The overall reaction is called aldol condensation. It is a nucleophilic addition-elimination reaction.
- The reaction can be given as,
\[\begin{array}{cc}
\ce{\underset{\text{Aldehyde}}{2R - CH2 - CHO} ->[aq.NaOH] R - CH2 - CH - CHR - CHO}\\
\phantom{.......................}|\\
\phantom{..........................}\ce{\underset{\text{Aldol}}{OH}}
\end{array}\]
\[\begin{array}{cc}
\ce{R - CH2 - CH - CHR - CHO ->[-H2O][warm] \underset{\text{α,β-Unsaturated aldehyde}}{R - CH = CR - CHO}}\\
|\phantom{.......................................}\\
\ce{\underset{Aldol}{OH}}\phantom{.....................................}
\end{array}\]
e.g.
- Ketones containing at least two α-hydrogen also undergo aldol condensation reaction and give an α,β-unsaturated ketone.
e.g.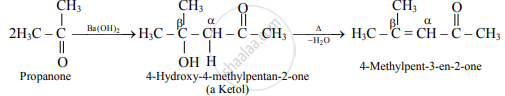
APPEARS IN
संबंधित प्रश्न
What is meant by the following term? Give an example of the reaction in the following case.
Aldol
Which of the following compounds would undergo aldol condensation, which the Cannizzaro reaction and which neither? Write the structures of the expected products of aldol condensation and Cannizzaro reaction.
- Methanal
- 2-Methylpentanal
- Benzaldehyde
- Benzophenone
- Cyclohexanone
- 1-Phenylpropanone
- Phenylacetaldehyde
- Butan-1-ol
- 2, 2-Dimethylbutanal
How will you convert ethanal into the following compound?
Butane-1, 3-diol
How will you convert ethanal into the following compound?
But-2-enal
Why is alpha (α) hydrogen of carbonyl compounds acidic in nature?
Write chemical equations of the following reaction :
Propanone is treated with dilute Ba (OH)2-.
Write chemical equations of the following reaction :
Benzoyl chloride is hydrogenated in the presence of `"Pd"/(BaSO_4)`
Write a chemical equation for the following reaction:
Propanone is treated with dilute Ba( OH)2.
Cannizaro’s reaction is not given by ______.
Which product is formed when the compound 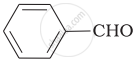 is treated with concentrated aqueous \[\ce{KOH}\] solution?
is treated with concentrated aqueous \[\ce{KOH}\] solution?
Compounds A and C in the following reaction are:
\[\ce{CH3CHO ->[(i) CH3MgBr][(ii) H2O] (A) ->[H2SO4, Δ] (B) ->[Hydroboration oxidation] (C)}\]
Which of the following conversions can be carried out by Clemmensen Reduction?
(i) Benzaldehyde into benzyl alcohol
(ii) Cyclohexanone into cyclohexane
(iii) Benzoyl chloride into benzaldehyde
(iv) Benzophenone into diphenyl methane
Why is there a large difference in the boiling points of butanal and butan-1-ol?
Assertion: Aromatic aldehydes and formaldehyde undergo Cannizaro reaction.
Reason: Aromatic aldehydes are almost as reactive as formaldehyde.
Cross aldol condensation occurs between
Which of the following gives aldol con~ensation reaction?
Convert the following:
Acetaldehyde to But-2-enal
Predict the reagent for carrying out the following transformations:
Ethanal to 3-hydroxy butanal
The major product of the following reaction is:
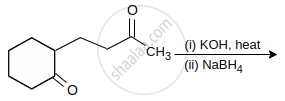
Which of the following does not give aldol condensation reaction?
Why is the α-hydrogens of aldehydes and ketones are acidic in nature?
Assertion (A): The final product in Aldol condensation is always α, β-unsaturated carbonyl compound.
Reason (R): α, β-unsaturated carbonyl compounds are stabilised due to conjugation.
When acetaldehyde is treated with dilute NaOH, the following reaction is observed.
\[\begin{array}{cc}
\ce{2CH3 - CHO ->[dil.NaOH] CH3 - CH - CH2 - CHO}\\
\phantom{...............}|\\
\phantom{.................}\ce{OH}
\end{array}\]
- What are the functional groups in the product?
- Can another product be formed during the same reaction? (Deduce the answer by doing atomic audit of reactant and product).
- Is this an addition reaction or condensation reaction?
Write a note on the aldol condensation reaction of acetaldehyde.
What is aldol condensation? Explain it with suitable examples.
