Advertisements
Advertisements
प्रश्न
Why is the α-hydrogens of aldehydes and ketones are acidic in nature?
Account for the following:
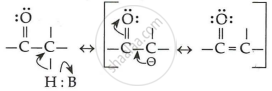
The alpha (α)-hydrogens of aldehydes and ketones are acidic in nature.
उत्तर
Because of the strong electron-withdrawing nature of the carbonyl groups and the resonance stabilisation of the conjugate base, aldehydes and ketones are acidic in nature.
| \[\begin{array}{cc} \ce{R}\phantom{..........}\\ \backslash\phantom{........}\\ \ce{C = O}\\ /\phantom{........}\\ \ce{H}\phantom{..........}\\ \end{array}\] |
\[\begin{array}{cc} \ce{R}\phantom{..........}\\ \backslash\phantom{........}\\ \ce{C = O}\\ /\phantom{........}\\ \ce{R}\phantom{..........}\\ \end{array}\] |
| Aldehyde | Ketone |
APPEARS IN
संबंधित प्रश्न
Write the products formed when CH3CHO reacts with the following reagents: CH3CHO in the presence of dilute NaOH
How will you convert ethanal into the following compound?
Butane-1, 3-diol
How will you convert ethanal into the following compound?
But-2-enal
Write structural formulas and names of four possible aldol condensation products from propanal and butanal. In each case, indicate which aldehyde acts as nucleophile and which as electrophile.
Complete the synthesis by giving missing starting material, reagent or product.
\[\begin{array}{cc}
\ce{C6H5CHO}\phantom{............}\\
\phantom{........}\ce{+\phantom{......}\ce{->[dil.NaOH][\Delta]}}\phantom{...}\\
\ce{CH3CH2CHO}\phantom{............}
\end{array}\]
What is substituted imine called?
Why is there a large difference in the boiling points of butanal and butan-1-ol?
What product will be formed on reaction of propanal with 2-methylpropanal in the presence of \[\ce{NaOH}\]? What products will be formed? Write the name of the reaction also.
Predict the reagent for carrying out the following transformations:
Ethanal to 3-hydroxy butanal
Which of the following does not give aldol condensation reaction?
