Advertisements
Advertisements
Questions
Why is the α-hydrogens of aldehydes and ketones are acidic in nature?
Account for the following:
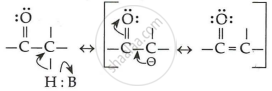
The alpha (α)-hydrogens of aldehydes and ketones are acidic in nature.
Solution
Because of the strong electron-withdrawing nature of the carbonyl groups and the resonance stabilisation of the conjugate base, aldehydes and ketones are acidic in nature.
| \[\begin{array}{cc} \ce{R}\phantom{..........}\\ \backslash\phantom{........}\\ \ce{C = O}\\ /\phantom{........}\\ \ce{H}\phantom{..........}\\ \end{array}\] |
\[\begin{array}{cc} \ce{R}\phantom{..........}\\ \backslash\phantom{........}\\ \ce{C = O}\\ /\phantom{........}\\ \ce{R}\phantom{..........}\\ \end{array}\] |
| Aldehyde | Ketone |
APPEARS IN
RELATED QUESTIONS
A compound 'A' of molecular formula C2H3OCl undergoes a series of reactions as shown below. Write the structures of A, B, C and D in the following reactions :
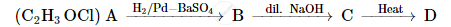
What is meant by the following term? Give an example of the reaction in the following case.
Aldol
How will you bring about the following conversion in not more than two steps?
Ethanol to 3-Hydroxybutanal
Write a chemical equation for the following reaction:
Propanone is treated with dilute Ba( OH)2.
What is substituted imine called?
Which product is formed when the compound 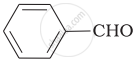 is treated with concentrated aqueous \[\ce{KOH}\] solution?
is treated with concentrated aqueous \[\ce{KOH}\] solution?
What product will be formed on reaction of propanal with 2-methylpropanal in the presence of \[\ce{NaOH}\]? What products will be formed? Write the name of the reaction also.
Which of the following gives aldol con~ensation reaction?
Convert the following:
Acetaldehyde to But-2-enal
What is aldol condensation? Explain it with suitable examples.
