Advertisements
Advertisements
प्रश्न
What is the action of following reagents on ethanoic acid?
P2O5/heat
उत्तर
Ethanoic acid on heating with strong dehydrating agent P2O5 forms acetic anhydride.
\[\begin{array}{cc}
\phantom{............}\ce{O}\phantom{........}\ce{O}\phantom{}\\
\phantom{............}||\phantom{........}||\phantom{}\\
\ce{\underset{\text{Ethanoic acid}}{2CH3COOH} ->[\ce{P2O5}/\Delta] \underset{\text{Acetic anhydride}}{CH3 _ C - O - C - CH3} + H2O}
\end{array}\]
संबंधित प्रश्न
Complete the synthesis by giving missing starting material, reagent or product.
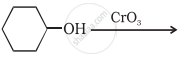
Complete the synthesis by giving missing starting material, reagent or product.

Complete the synthesis by giving missing starting material, reagent or product.
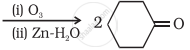
Write a note on ‘aldol condensation’.
What is the action of phenyl hydrazine on propanone?
What is the action of Zn – Hg / conc. HCl on propanone?
What is the action of Sodium bisulphite on propanone?
Write only reactions for the preparation of benzophenone from benzonitrile.
Write chemical equation for the following:
Gatterman - Koch formylation
Write the IUPAC name of the follow
\[\begin{array}\ce{\phantom{--..}O}\\\ce{\phantom{--..}||}\\ \ce{CH2=CH-C-CH3}\end{array}\]
Complete the following reaction:
\[\ce{(C6H5CH2)2Cd + 2CH3COCI}\]
Answer the following in one sentence.
Predict the product of the following reaction:
\[\ce{CH3 - CH2 - COOCH3->[i) AIH(i-Bu)_2][ii) H3O+]}\]?
What is the action of following reagents on ethanoic acid?
SOCl2/heat
Draw structure of salicylaldehyde.
The formation of aldehyde from an alkyl cyanide using SnCl2/conc.HCl is known as ____________.
In the formation of an aldol, α-carbon atom of one aldehyde molecule attaches to ____________ of another aldehyde molecule.
Catalyst used in Rosenmund reduction is ____________.
An acylchloride is hydrogenated over catalyst palladium on barium sulphate to form an aldehyde. This reaction is called as ______.
Which of the following represents Etard's reaction?
Rosenmund reduction of ____________ yields benzaldehyde.
Identify the compound obtained by side chain chlorination of toluene and hydrolysed further.
Identify 'A' and 'B' respectively in following reaction.
\[\ce{Toluene ->[Cl2][hv] A ->[H2O][\Delta] B}\]
The product of the reaction between dimethylcadmium and acetyl chloride is ____________.
Identify the compound A and Bin following reaction.
\[\ce{CH3Cl ->[KCN(alc)][\Delta] A ->[2H2O][HCl] B + NH4Cl}\]
A reaction in which chromyl chloride in presence of CS2 converts methyl group of toluene to aldehydic group on acid hydrolysis is called _________.
Which among the following compounds is obtained when benzene is treated with CO and HCl in presence of catalyst anhydrous AlCl3 and Cu2Cl2 under high pressure?
Which of the following compounds does NOT undergo Friedel-Crafts reaction?
What is the final product obtained when benzonitrile react with phenyl magnesium bromide in equimolar proportion?
Which of the following is obtained by hydrogenation of benzoyl chloride in presence of Pd on BaSO4?
Given below are two statements labelled as Assertion (A) and Reason (R).
Assertion (A): SO2 is reducing while TeO2 is an oxidising agent.
Reason (R): Reducing property of dioxide decreases from SO2 to TeO2.
Select the most appropriate answer from the options given below:
How ketones are prepared from nitriles?
Give the reactions involved in the Etard's reaction.
Two isomers 'A' and 'B' with molecular formula C4H8 give different products on oxidation with KMnO4 in acidic medium. Isomer 'A' on reaction with KMnO4/H+ results in the effervescence of gas and gives ketone. The compound 'A' is:
Which reagent is used in Etard reaction?
What is the action of hydrogen cyanide on the following?
Benzaldehyde
Write Stephen reaction for the preparation of aldehyde.
How butanone is prepared from alcohol?
How are the following compound obtained by hydration of alkenes?
Isopropyl alcohol
Explain Rosenmund reduction reaction.
How acetone is prepared from nitriles using Grignard reagent?
Write preparation of propanone by using ethanoyl chloride and dimethyl cadmium.
