Advertisements
Advertisements
प्रश्न
Give reasons for the following:
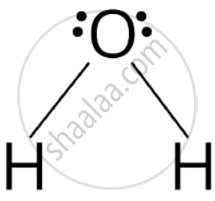
Water molecule has bent structure whereas carbon dioxide molecule is linear.
टिप्पणी लिखिए
उत्तर
Due to presence of two lone pairs of electrons on oxygen atom in \[\ce{HiO}\] the repulsion between Ip-lp is more. \[\ce{CO2}\] undergoes sp hybridization resulting in linear shape \[\ce{(O = C = O)}\] while \[\ce{H2O}\] undergoes sp3 hybridisation resulting in distorted tetrahedral or bent structure.
shaalaa.com
क्या इस प्रश्न या उत्तर में कोई त्रुटि है?
अध्याय 4: Chemical Bonding and Molecular Structure - Multiple Choice Questions (Type - I) [पृष्ठ ४५]
