Advertisements
Advertisements
प्रश्न
Give simple chemical tests to distinguish between the following pairs of compounds :
Ethanol and phenol
उत्तर १
| Test | Ethanol | Phenol |
| Coupling reaction | Negative test |  |
| Iodoform test |  |
Negative test |
उत्तर २
Ethanol and phenol can be chemically distinguished by Iodoform test
\[\ce{C2H5OH->[I2][NaOH][CH5CHo]->\underset{\text{Yellow precipitate}}{CHI3} v}\]

APPEARS IN
संबंधित प्रश्न
Give reasons for the following : Boiling point of ethanol is higher in comparison to methoxymethane.
Explain why propanol has higher boiling point than that of the hydrocarbon, butane?
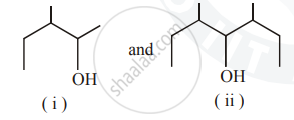
Identify the chiral molecule in the following pair :
Write the structures of A, B and C in the following reactions :
\[ C_6 H_5 {NO}_2 \to^{Sn/HCI} A \to^{{NaNO}_2 /HCI}_{273 K} B \to^{H_2 O}_∆ C\]
Alcohols of low molecular weight are _____________.
Assertion: Boiling points of alcohols and ethers are high.
Reason: They can form intermolecular hydrogen-bonding.
How are the following conversion carried out?
Methyl magnesium bromide →2-Methylpropan-2-ol.
What is esterifications? How is an ester obtained from alcohol or phenol?
Convert the following:
acetaldehyde to isopropyl alcohol.
How are the following conversions carried out?
\[\ce{Methyl magnesium bromide ->2-Methylpropan-2-ol}\]
