Advertisements
Advertisements
प्रश्न
Give the construction and working of photo emissive cell.
उत्तर
Photo emissive cell: Its working depends on the electron emission from a metal cathode due to irradiation of light or other radiations.
Construction:
1. It consists of an evacuated glass or quartz bulb in which two metallic electrodes – that is, a cathode and an anode are fixed.
2. The cathode C is semi-cylindrical in shape and is coated with a photo sensitive material. The anode A is a thin rod or wire kept along the axis of the semi-cylindrical cathode.
3. A potential difference is applied between the anode and the cathode through a galvanometer G.
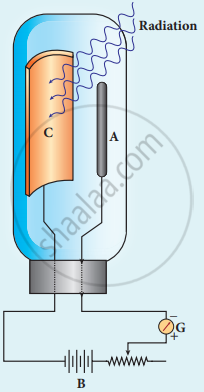
Construction of photo cell
Working:
When the cathode is irradiated with suitable radiation, electrons are emitted from it. These electrons are attracted by the anode and hence a current is produced which is measured by the galvanometer.
For a given cathode, the magnitude of the current depends on
- the intensity to incident radiation and
- the potential difference between anode and cathode.
APPEARS IN
संबंधित प्रश्न
The threshold wavelength for a metal surface whose photoelectric work function is 3.313 eV is __________.
A light of wavelength 500 nm is incident on a sensitive metal plate of photoelectric work function 1.235 eV. The kinetic energy of the photoelectrons emitted is (Take h = 6.6 × 10–34 Js)
The work functions for metals A, B and C are 1.92 eV, 2.0 eV and 5.0 eV respectively. The metal/metals which will emit photoelectrons for a radiation of wavelength 4100Å is/are
What is a surface barrier?
Mention the two features of x-ray spectra, not explained by classical electromagnetic theory.
Explain the effect of potential difference on photoelectric current.
Explain the quantum concept of light.
Give the applications photocell.
Calculate the maximum kinetic energy and maximum velocity of the photoelectrons emitted when the stopping potential is 81 V for the photoelectric emission experiment.
UV light of wavelength 1800 Å is incident on a lithium surface whose threshold wavelength is 4965 Å. Determine the maximum energy of the electron emitted.
