Advertisements
Advertisements
प्रश्न
Give the steps involved in the extraction of metals of low and medium reactivity from their respective sulphide ores.
उत्तर
In the extraction of metal from their sulphide ores, the ore is first kept for the process of roasting in which it is heated in the presence of air below its melting point to convert the sulphide ore to its oxide form.
For example, Cinnabar is heated in the air which mercuric sulphide gets oxidised to produce mercuric oxide.
`2"HgS" + 3"O"_2 -> 2"HgO" + 2"SO"_2`
Mercuric oxide is further reduced by heating to get pure mercury.
`2"HgO" -> 2"Hg" + "O"_2`
In the case of zinc which is a metal of medium reactivity and its ore is zinc blende.
Zinc blend is roasted to get zinc oxide.
`2"Zns" + 3"O"_2 -> 2"ZnO" + 2"SO"_2`
When zinc oxide is reduced in the presence of coke, pure zinc metal is produced.
`"ZnO" + "C" -> "Zn" + "CO"`
APPEARS IN
संबंधित प्रश्न
What do you observe when a few pieces of iron are dropped in a blue solution of copper sulphate?
What do you observe when ferrous sulphate solution is added to an aqueous solution of sodium hydroxide.
How will you obtain Silver chloride from silver nitrate.
Also give balanced equations for the reactions
What do you observe when Magnesium ribbon is burnt in oxygen.
State what is meant by the ‘reactivity series of metals’
Complete the statement by filling in the blank with the correct word:
The metal which reacts with steam and the reaction is reversible is ________.
Observe the following diagram and identify the type of reaction and write observation.
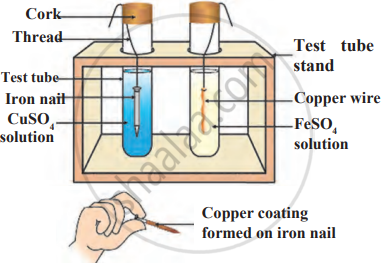
Which among the following alloys contain mercury as one of its constituents?
Explain the following
- Reactivity of Al decreases if it is dipped in HNO3
- Carbon cannot reduce the oxides of Na or Mg
- NaCl is not a conductor of electricity in solid state whereas it does conduct electricity in aqueous solution as well as in molten state
- Iron articles are galvanised.
- Metals like Na, K, Ca and Mg are never found in their free state in nature.
An element A burns with golden flame in air. It reacts with another element B, atomic number 17 to give a product C. An aqueous solution of product C on electrolysis gives a compound D and liberates hydrogen. Identify A, B, C and D. Also write down the equations for the reactions involved.
