Advertisements
Advertisements
प्रश्न
How are 1 - nitropropane, 2-nitropropane and 2-methyl 2- nitropropane are distinguished from each other using nitrous acid?
उत्तर
1-Nitropropane reacts with nitrous acid to form blue-coloured nitrosonitroalkanes which dissolve in NaOH to give red solution.

2-Nitropropane reacts with nitrous acid to form blue-coloured nitrosonitroalkanes which are further insoluble in NaOH because of the absence of the alpha hydrogen atom.
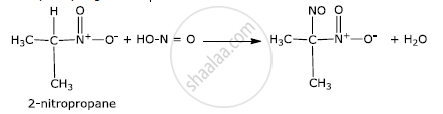
2-Methyl2-nitropropane does not react with nitrous acid because it has no alpha-hydrogen atom.
APPEARS IN
संबंधित प्रश्न
Primary and secondary nitroalcanes containing α - H atom show property of -
- chain isomerism
- tautomerism
- optical isomerism
- geometrical isomerism
Distinguish between the following pairs of compounds: Aniline and N-methylaniline
Describe a method for the identification of primary, secondary and tertiary amines. Also write chemical equations of the reactions involved.
Give plausible explanation for each of the following:
Why do primary amines have higher boiling point than tertiary amines?
What is the action of nitrous acid on tertiary nitroalkane
Which of the following reactions will not give a primary amine?
On heating an aliphatic primary amine with chloroform and ethanolic potassium hydroxide, the organic compound formed is ______.
Primary, secondary and tertiary amines may be separated by using ______.
Identify the compound that will react with Hinsberg’s reagent to give a solid which dissolves in alkali.
Arrange the following in the increasing order of their boiling points:
A: Butanamine
B: N, N-Dimethylethanamine
C: N- Etthylethanaminamine
Given below are two statements labelled as Assertion (A) and Reason (R).
Assertion (A): Tertiary amines are more basic than corresponding secondary and primary amines in gaseous state.
Reason (R): Tertiary amines have three alkyl groups which cause +I effect.
Select the most appropriate answer from the options given below:
Compound A is converted to B on reaction with CHCl3 and KOH. The compound B is toxic and can be decomposed by C. A, B and C respectively are ______.
Isocyanide reaction involves the intermediate formation of ______.
Which of the following is not a correct statement for primary aliphatic amines?
Match List I with List II.
| List I | List II | ||
| (A) | Benzenesulphonyl chloride | (I) | Test for primary amines |
| (B) | Hoffmann bromamide reaction | (II) | Anti Saytzeff |
| (C) | Carbylamine reaction | (III) | Hinsberg reagent |
| (D) | Hoffmann orientation | (IV) | Known reaction of Isocyanates |
Choose the correct answer from the options given below:
A compound 'X' on treatment with Br2/NaOH, provided C3H9N, which gives positive carbylamine test. Compound 'X' is ______.
Give reasons:
(CH3)2NH is more basic than (CH3)3N in an aqueous solution.
Explain why (CH3)2NH is more basic than (CH3)3N in aqueous solution.
In the reaction \[\ce{C6H5NH2 + CHCl3 + 3KOH -> A + 3B + 3C}\] the product A is:
Carbylamine test is the distinguishing test for ______.
Among dimethylamine (pKb = 3.27) and diethylamine (pKb = 3.0), which one is more basic?
