Advertisements
Advertisements
प्रश्न
How does Huckel rule help to decide the aromatic character of a compound?
उत्तर
In 1865, August Kekule suggested that benzene consists of a cyclic planar structure of six carbon with alternate single and double bonds.
There were two objections:
- Benzene forms only one ortho disubstituted products whereas the Kekule’s structure predicts two o-di substituted products as shown below.

- Kekule’s structure failed to explain why benzene with three double bonds did not give addition reactions like other alkenes.To overcome this objection, Kekule suggested that benzene was mixture of two forms (1 and 2) which are in rapid equilibrium.

- Resonance description of benzene:
The phenomenon in which two or more structures can be written for a substance which has an identical position of atoms is called resonance. The actual structure of the molecule is said to be resonance hybrid of various possible alternative structures. In benzene, Kekule’s structures I & II represented the resonance structure, and structure III is the resonance hybrid of structure I & II.
The structures 1 and 2 exist only in theory. The actual structure of benzene is the hybrid of two hypothetical resonance structures.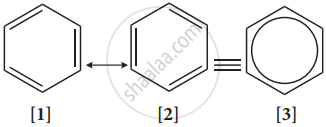
- Spectroscopic measurements:
Spectroscopic measurements show that benzene is planar and all of its carbon-carbon bonds are of equal length 1.40 Å. This value lies between carbon-carbon single bond length 1.54 Å and carbon-carbon double bond length 1.34 Å. - Molecular orbital structure:
The structure of benzene is best de¬scribed in terms of the molecular orbital theory. All the six carbon atoms of benzene are sp2 hybridized. Six sp2 hybrid orbitals of carbon linearly overlap with six 1 s orbitals of hydrogen atoms to form six C – H sigma bonds. Overlap between the remaining sp2 hybrid orbitals of carbon forms six C – C sigma bonds.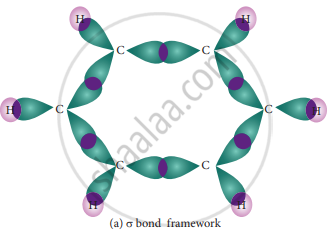
- Formation of Sigma bond in benzene-
All the σ bonds in benzene lie in one plane with a bond angle of 120°. Each carbon atom in benzene possess a un hybridized p-orbital containing one electron. The lateral overlap of their p-orbital produces 3 π- bond. The six electrons of the p-orbitals cover all the six carbon atoms and are said to be delocalized. Due to delocalization, strong π-bond is formed which makes the molecule stable. Hence unlike alkenes and alkynes benzene undergoes substitution reactions rather addition reactions under normal conditions.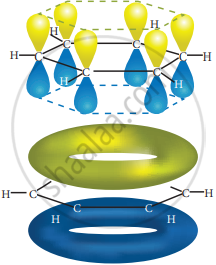
All carbon atoms have the delocalized π MO is formed by p-orbitais the overlap of six p-orbitals. - Representation of benzene:
Hence, there are three ways in which benzene can be represented.
Expanded form-
Kekule structure-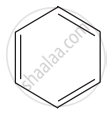
Short hand representation
APPEARS IN
संबंधित प्रश्न
Write the balanced chemical reaction to get benzene from Phenol.
Major product of the below mentioned reaction is, \[\ce{(CH3 )2 C = CH2 ->[ICI]}\]
Which of the following compounds will not undergo Friedal – crafts reaction easily ? (NEET)
Which of the following is aliphatic saturated hydrocarbon?
Suggest the route for the preparation of the following from benzene.
3 – chloro nitrobenzene
What happens when ethylene is passed through cold dilute alkaline potassium permanganate.
A compound with molecular formula C4H4O has all the four carbon atom and the oxygen atom in the ring. It also has two carbon-carbon double bonds. The compound is ____________.
Dow's process is used for the synthesis of an aromatic compound (X). Identify X.
Arenes on treatments with chlorine in the presence of ferric chloride as a catalyst undergo what type of reaction?
Read the following reaction and answer the questions given below.
\[\begin{array}{cc}
\phantom{..............................}\ce{CH3}\\
\phantom{...........................}|\\
\ce{CH3 - C = CH2 + HBr ->[Benzoyl][peroxide] H3C - CH - CH2Br}\\
|\phantom{....................................}\\
\ce{CH3}\phantom{.................................}
\end{array}\]
- Write the IUPAC name of the product.
- State the rule that governs the formation of this product.
