Advertisements
Advertisements
प्रश्न
How is methane prepared in the laboratory?
उत्तर
Reactants: Sodium ethanoate (sodium acetate) and soda lime.
Since sodium hydroxide is deliquescent, soda lime (a mixture of sodium hydroxide and calcium oxide) is used, which is in the form of a white porous mass or granules.
Procedure: A mixture of sodium ethanoate (sodium acetate) and soda lime is taken in a hard glass test tube (as shown in Fig.) and heated over a Bunsen burner.
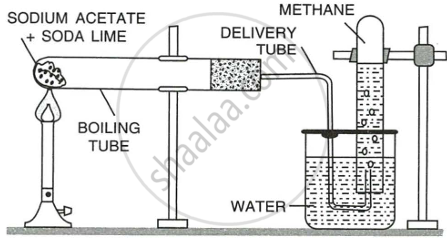
Reaction:
\[\ce{\underset{acetate}{\underset{sodium}{CH3COONa}} + \underset{hydroxide}{\underset{sodium}{NaOH}} ->[CaO,300^\circ C] \underset{carbonate}{\underset{Sodium}{Na2CO3}} + \underset{Methane}{CH4}}\]
Collection: The gas evolved is collected by downward displacement of water since it is slightly soluble in water and is lighter than air.
APPEARS IN
संबंधित प्रश्न
Name the following:
A hydrocarbon which contributes towards the greenhouse effect.
Match the options A to E with the statements
| A | Alkyne | 1 | No. of molecules in 22.4 dm-3 of carbon dioxide at STP |
| B | Alkane | 2 | An element with the electronic configuration 2,8,8,3 |
| C | Iron | 3 | CnH2n + 2 |
| D | 6.023 × 1023 | 4 | CnH2n − 2 |
| E | Metal | 5 | The metal which forms two types of ions |
Write the IUPAC and common names of these isomers
Name the hydrocarbon which is known as paraffin.
Write the names and the formula of the products formed when
methane Reacts with chlorine Write the chemical equations
Write the names and the formula of the products formed when ethane reacts with bromine. Write the chemical equations.
What is an alkyl group?
Write a balanced chemical equation for the preparation of :
Ethyne using calcium carbide
Identify the statement that is incorrect about alkanes.
Which type of reaction will ethane undergo?
