Advertisements
Advertisements
प्रश्न
How is the following metallic oxide reduced? Write the equation:
Zinc oxide
उत्तर
It is reduced by a carbon reduction process using a suitable reducing agent, carbon (C).
ZnO and Coke are heated in retort by producer gas (CO + N2) to get zinc spelter.
\[\ce{ZnO(s) + C(s) ->[1400^\circ C] \underset{(Zinc spelter)}{Zn(s)} + CO(g)}\]
APPEARS IN
संबंधित प्रश्न
Name the compound added to pure alumina to lower the fusion temperature during the electrolytic reduction of alumina.
Explain with reason:
In the electrolytic reduction of alumina, the graphite anode is gradually consumed.
Give reason for the following:
Carbon anodes are used in the electrolytic extraction of aluminium.
Give equation for the following conversion: Ferric oxide to iron.
Give equation for the following conversion: Aluminium hydroxide to aluminium oxide.
Give the chemical name and formula of 'cryolite'
Name a non-metallic element which forms acidic and neutral oxides
Name a non-metallic element which has a metallic lustre
Write balanced equation for the following reaction:
Reduction of copper oxide by hydrogen.
The following sketch represents the electroplating of an Iron cup with Nickel metal.
Study the diagram and answer the following questions:
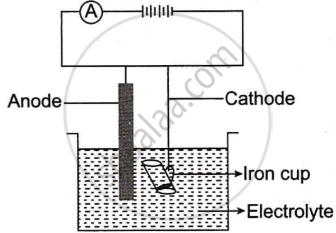
- During electroplating, the iron cup is placed at the cathode. Why?
- Name the ion that must be present in the electrolyte.
- State one condition that is necessary to ensure that the deposit is smooth, firm and even.
- Write the reaction taking place at the cathode.
- What change would you observe at the anode?
