Advertisements
Advertisements
प्रश्न
How is the following metallic oxide reduced? Write the equation:
Zinc oxide
उत्तर
It is reduced by a carbon reduction process using a suitable reducing agent, carbon (C).
ZnO and Coke are heated in retort by producer gas (CO + N2) to get zinc spelter.
\[\ce{ZnO(s) + C(s) ->[1400^\circ C] \underset{(Zinc spelter)}{Zn(s)} + CO(g)}\]
APPEARS IN
संबंधित प्रश्न
Give appropriate scientific reasons for Zinc oxide can be reduced to zinc metal by using
carbon, but aluminium oxide cannot be reduced by a reducing agent
Give equation for the following conversion: Ferric oxide to iron.
Name a non-metallic element which forms acidic and neutral oxides
Write balanced equation for the following reaction:
Reduction of copper oxide by hydrogen.
Write balanced equation for the following reaction:
Reduction of lead (II) oxide by carbon.
Name the following :
A metal oxide that can be reduced by hydrogen
Complete the following by selecting the correct option from the choices given :
The diavalent metal whose oxide is reduced to metal by electrolysis of its fused salt is ________
Name two metallic oxides which cannot be reduced by carbon, carbon monoxide or hydrogen.
Write a balanced equation for the reduction of copper (ll) oxide by hydrogen.
The following sketch represents the electroplating of an Iron cup with Nickel metal.
Study the diagram and answer the following questions:
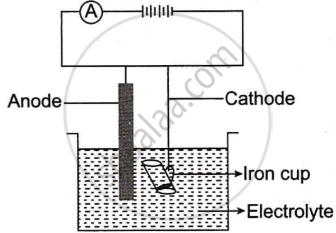
- During electroplating, the iron cup is placed at the cathode. Why?
- Name the ion that must be present in the electrolyte.
- State one condition that is necessary to ensure that the deposit is smooth, firm and even.
- Write the reaction taking place at the cathode.
- What change would you observe at the anode?
