Advertisements
Advertisements
प्रश्न
How will you convert ethyl chloride into n – butane?
उत्तर
\[\ce{\underset{\text{}Ethyl chloride}{C2H5Cl + 2Na +}Cl - C2H5 ->[-2NaBr][dry ether] CH3 - \underset{\text{n - butane}}{CH2 - CH2 - CH3}}\]
APPEARS IN
संबंधित प्रश्न
Identify the main product of the reaction.
\[\begin{array}{cc}
\ce{CH3 - CH - CH3 ->[{60%}H2SO4][373 K]}\\
|\phantom{............}\\\ce{OH}\phantom{..........}
\end{array}\] _______.
Identify A, B, C in the following reaction sequence:
\[\ce{CH3 - CH = CH2 ->[Br2/CCl4][room temperature] A ->[Zn] B ->[dil. alkaline][KMnO4] C}\]
Name two reagents used for acylation of benzene.
Major product of the below mentioned reaction is, \[\ce{(CH3 )2 C = CH2 ->[ICI]}\]
Which of the following compounds will not undergo Friedal – crafts reaction easily ? (NEET)
Identify the compound 'Z' in the following reaction
\[\ce{C2H6O ->[Al2O3][623 K] X ->[O3] Y ->[Zn/H2O] (Z)}\]
Describe the mechanism of Nitration of benzene.
Identify the X and Y in the following reactions.
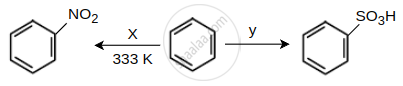
Read the following reaction and answer the questions given below.
\[\begin{array}{cc}
\phantom{..............................}\ce{CH3}\\
\phantom{...........................}|\\
\ce{CH3 - C = CH2 + HBr ->[Benzoyl][peroxide] H3C - CH - CH2Br}\\
|\phantom{....................................}\\
\ce{CH3}\phantom{.................................}
\end{array}\]
- Write the IUPAC name of the product.
- State the rule that governs the formation of this product.
Read the following reaction and answer the questions given below.
\[\begin{array}{cc}
\phantom{..........................}\ce{CH3}\\\phantom{........................}|\\\ce{CH3 - C = CH2 + HBr ->[benzoyl][peroxide] CH3 - CH - CH2Br}\\
|\phantom{......................................}\\
\ce{CH3}\phantom{....................................}\end{array}\]
- Write the IUPAC name of the product.
- State the rule that governs the formation of this product.
