Advertisements
Advertisements
प्रश्न
Identify A, B, C in the following reaction sequence:
\[\ce{CH3 - CH = CH2 ->[Br2/CCl4][room temperature] A ->[Zn] B ->[dil. alkaline][KMnO4] C}\]
उत्तर
\[\begin{array}{cc}
\ce{H\phantom{....}H\phantom{...............}}\\
|\phantom{.....}|\phantom{...............}\\
\ce{CH3\underset{Propene}{- CH =} CH2 ->[Br2/CCl4][room temperature] CH3 - C - C - H ->[Zn] CH3 \underset{\overset{Propene}{(B)}}{- CH = C}H2 ->[dil.alkaline][KMnO4] CH3 - CH - CH2}\\
\phantom{........................................}|\phantom{.....}|\phantom{..............................................}|\phantom{......}|\phantom{..}\\
\ce{\phantom{.......................................}\underset{\overset{1,2-Dibromopropane}{(A)}}{Br\phantom{...} Br}\phantom{...........................................}\underset{\overset{Propane-1,2-diol}{(C)}}{OH\phantom{...}OH}}\end{array}\]
APPEARS IN
संबंधित प्रश्न
Predict the possible product of the following reaction.
Chlorination of nitrobenzene
Identify the main product of the reaction.
\[\begin{array}{cc}
\ce{CH3 - CH - CH3 ->[{60%}H2SO4][373 K]}\\
|\phantom{............}\\\ce{OH}\phantom{..........}
\end{array}\] _______.
Identify the main product of the reaction:
\[\ce{HC ≡ C - CH3 ->[H2][Pd-C/quinoline]}\] _______.
Identify the main product of the reaction:
\[\ce{H - C ≡ C - H + H - O ->[{40%} H2SO4][{1%} HgSO4]}\] _____.
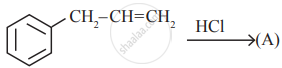 is
is
An alkane is obtained by decarboxylation of sodium propionate. Same alkane can be prepared by ______.
Which of the following is aliphatic saturated hydrocarbon?
2 – butyne on chlorination gives ______.
Give IUPAC name for the following compound.
\[\ce{CH ≡ C – C ≡ C – C ≡ CH}\]
Write short notes on ortho, para directors in aromatic electrophilic substitution reactions.
Suggest a simple chemical test to distinguish propane and propene.
Suggest the route for the preparation of the following from benzene.
3 – chloro nitrobenzene
Suggest the route for the preparation of the following from benzene.
4 – chlorotoluene
Suggest the route for the preparation of the following from benzene.
Bromo benzene
Write the structure of the following alkanes.
2, 3 – Dimethyl – 6 – (2 – methyl propyl) decane
Write the structure of the following alkanes.
5 – (2 – Ethyl butyl) – 3, 3 – dimethyldecane
Write the structure of the following alkanes.
5 – (1, 2 – Dimethyl propyl) – 2 – methylnonane
How will you prepare propane from a sodium salt of fatty acid?
Which of the following is NOT a hetero-aromatic compound?
−Cl group is ____________.
Which of the following is INCORRECT for aromatic hydrocarbons?
α-butylene when subjected to hydroboration oxidation reaction, yields ____________.
Which of the following molecules has shortest C - C bond length?
Which of the following reagents can bring about following conversion?
\[\ce{But-1-ene -> Butan-2-ol}\]
Which of the following compounds is NOT obtained in thermal decomposition of propane in absence of air?
Which of the following compounds on bromination yields ![]() ?
?
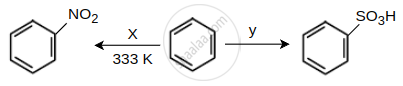
Identify the X and Y in the following reactions.
Read the following reaction and answer the questions given below.
\[\begin{array}{cc}
\phantom{...............................}\ce{CH3}\\
\phantom{............................}|\\
\ce{CH3 - C = CH2 + HBr ->[benzoyl][peroxide]CH3 - CH - CH2Br}\\
|\phantom{....................................}\\
\ce{CH3}\phantom{..................................}
\end{array}\]
- Write the IUPAC name of the product.
- State the rule that governs the formation of this product.
Read the following reaction and answer the questions given below.
\[\begin{array}{cc}
\phantom{..............................}\ce{CH3}\\
\phantom{............................}|\\
\ce{CH3 - C = CH2 + HBr ->[benzoyl][peroxide]CH3 - CH - CH2Br}\\
|\phantom{....................................}\\
\ce{CH3}\phantom{.................................}
\end{array}\]
- Write IUPAC name of the product.
- State the rule that governs formation of this product.
