Advertisements
Online Mock Tests
Chapters
2: Introduction to Analytical Chemistry
3: Basic Analytical Techniques
4: Structure of Atom
5: Chemical Bonding
6: Redox Reactions
7: Modern Periodic Table
8: Elements of Group 1 and 2
9: Elements of Group 13, 14 and 15
10: States of Matter
11: Adsorption and Colloids
12: Chemical Equilibrium
13: Nuclear Chemistry and Radioactivity
14: Basic Principles of Organic Chemistry
▶ 15: Hydrocarbons
16: Chemistry in Everyday Life
![Balbharati solutions for Chemistry [English] 11 Standard chapter 15 - Hydrocarbons Balbharati solutions for Chemistry [English] 11 Standard chapter 15 - Hydrocarbons - Shaalaa.com](/images/chemistry-english-11-standard_6:1ddd95908cb04440a83d42566c3337de.jpg)
Advertisements
Solutions for Chapter 15: Hydrocarbons
Below listed, you can find solutions for Chapter 15 of Maharashtra State Board Balbharati for Chemistry [English] 11 Standard.
Balbharati solutions for Chemistry [English] 11 Standard 15 Hydrocarbons Exercises [Pages 259 - 260]
Choose the correct option.
Which of the following compound has the highest boiling point?
n-pentane
iso-butane
butane
neopentane
Choose the correct option.
Acidic hydrogen is present in _______.
acetylene
ethane
ethylene
dimethyl acetylene
Choose the correct option.
Identify 'A' in the following reaction:
\[\begin{array}{cc}
\ce{CH3 - C = CH2 ->[A] CH3 - C - CH3 + CO2 + H2O}\\|\phantom{...................}||\phantom{..............}\\
\ce{CH3}\phantom{................}\ce{O}\phantom{..............}
\end{array}\]
KMnO4/H+
alkaline KMnO4
dil. H2SO4/1% HgSO4
NaOH/H2O2
Choose the correct option.
The major product of chlorination of ethyl benzene is _______.
m-chlorethyl benzene
p - chloroethyl benzene
chlorobenzene
o - chloroethylbenzene
Choose the correct option.
1-Chloropropane on treatment with alc. KOH produces _______.
propane
propene
propyne
propyl alcohol
Name the following:
The type of hydrocarbon that is used as a lubricant.
Name the following:
Alkene used in the manufacture of polythene bags.
Name the following:
The hydrocarbon said to possess carcinogenic property.
Name the following:
What are the main natural sources of alkane?
Arrange the three isomers of alkanes with molecular formula C5H12 in increasing order of boiling points and write their IUPAC names.
Write IUPAC names of the products obtained by the reaction of cold concentrated sulphuric acid followed by water with the following compounds.
Propene
Write IUPAC names of the products obtained by the reaction of cold concentrated sulphuric acid followed by water with the following compound.
but-l-ene
Write a balanced chemical reaction for the preparation of ethane from Ethyl bromide.
Write a balanced chemical reaction for the preparation of ethane from Ethyl magnesium iodide.
How many monochlorination product is possible for 2-methylpropane? Draw their structures and write their IUPAC names.
How many monochlorination product is possible for 2-methylbutane? Draw their structures and write their IUPAC names.
Write all the possible products for pyrolysis of butane.
Which of the following will exhibit geometrical isomerism?
\[\begin{array}{cc}\ce{CH3 - Ch2 - C - Ch3}\\
\phantom{.......}||\\\phantom{.........}\ce{CH2}
\end{array}\](CH3)2C = CH2
\[\begin{array}{cc}
\ce{CH3 - C = C - CH3}\\|\phantom{....}|\\
\phantom{...}\ce{C2H5}\phantom{.}\ce{C2H5}
\end{array}\]
What is the action of following on ethyl iodide?
alc. KOH
What is the action of following on ethyl iodide?
Zn, HCl
An alkene ‘A’ on ozonolysis gives 2 moles of ethanal. Write the structure and IUPAC name of ‘A’.
Acetone and acetaldehyde are the ozonolysis products of an alkene. Write the structural formula of an alkene and give IUPAC name of it.
Write the reaction to convert propene to n-propyl alcohol.
Write the reaction to convert propene to isopropyl alcohol.
What is the action of following on but-2-ene?
dil. alkaline KMnO4
What is the action of following on but-2-ene?
acidic KMnO4
Complete the following reaction sequence:
\[\ce{HC ≡ CH ->[LiNH2] A ->[CH3CH2Cl] B ->[HBr] C}\]
Comment on the acidic nature of B.
Write the balanced chemical reaction to get benzene from Sodium benzoate.
Write the balanced chemical reaction to get benzene from Phenol.
Predict the possible product of the following reaction.
Chlorination of nitrobenzene
Predict the possible product of the following reaction.
sulphonation of chlorobenzene
Predict the possible product of the following reaction.
bromination of phenol
Predict the possible product of the following reaction.
nitration of toluene
Identify the main product of the reaction:
\[\ce{CH3 - CH2 - CH3 ->[O2][\Delta]}\] ______.
Identify the main product of the reaction.
\[\begin{array}{cc}
\ce{CH3 - CH - CH3 ->[{60%}H2SO4][373 K]}\\
|\phantom{............}\\\ce{OH}\phantom{..........}
\end{array}\] _______.
Identify the main product of the reaction:
\[\ce{HC ≡ C - CH3 ->[H2][Pd-C/quinoline]}\] _______.
Identify the main product of the reaction:
\[\ce{H - C ≡ C - H + H - O ->[{40%} H2SO4][{1%} HgSO4]}\] _____.
Read the following reaction and answer the questions given below.
\[\begin{array}{cc}
\phantom{..............................}\ce{CH3}\\\phantom{...........................}|\\\ce{CH3 - C = CH2 + HBr ->[benzoyl][peroxide] CH3 - CH - CH2Br}\\
|\phantom{....................................}\\
\ce{CH3}\phantom{.................................}\end{array}\]
- Write the IUPAC name of the product.
- State the rule that governs the formation of this product.
Identify A, B, C in the following reaction sequence:
\[\ce{CH3 - CH = CH2 ->[Br2/CCl4][room temperature] A ->[Zn] B ->[dil. alkaline][KMnO4] C}\]
Identify giving reason whether the following compound is aromatic or not.
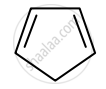
Identify giving reason whether the following compound is aromatic or not.

Identify giving reason whether the following compound is aromatic or not.
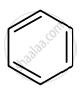
Identify giving reason whether the following compound is aromatic or not.
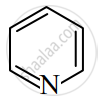
Name two reagents used for acylation of benzene.
Read the following reaction and answer the questions given below:
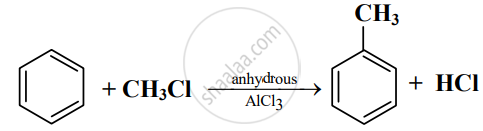
- Write the name of the reaction.
- Identify the electrophile in it.
- How is this electrophile generated?
Solutions for 15: Hydrocarbons
![Balbharati solutions for Chemistry [English] 11 Standard chapter 15 - Hydrocarbons Balbharati solutions for Chemistry [English] 11 Standard chapter 15 - Hydrocarbons - Shaalaa.com](/images/chemistry-english-11-standard_6:1ddd95908cb04440a83d42566c3337de.jpg)
Balbharati solutions for Chemistry [English] 11 Standard chapter 15 - Hydrocarbons
Shaalaa.com has the Maharashtra State Board Mathematics Chemistry [English] 11 Standard Maharashtra State Board solutions in a manner that help students grasp basic concepts better and faster. The detailed, step-by-step solutions will help you understand the concepts better and clarify any confusion. Balbharati solutions for Mathematics Chemistry [English] 11 Standard Maharashtra State Board 15 (Hydrocarbons) include all questions with answers and detailed explanations. This will clear students' doubts about questions and improve their application skills while preparing for board exams.
Further, we at Shaalaa.com provide such solutions so students can prepare for written exams. Balbharati textbook solutions can be a core help for self-study and provide excellent self-help guidance for students.
Concepts covered in Chemistry [English] 11 Standard chapter 15 Hydrocarbons are Alkanes, Alkenes, Alkynes, Aromatic Hydrocarbons.
Using Balbharati Chemistry [English] 11 Standard solutions Hydrocarbons exercise by students is an easy way to prepare for the exams, as they involve solutions arranged chapter-wise and also page-wise. The questions involved in Balbharati Solutions are essential questions that can be asked in the final exam. Maximum Maharashtra State Board Chemistry [English] 11 Standard students prefer Balbharati Textbook Solutions to score more in exams.
Get the free view of Chapter 15, Hydrocarbons Chemistry [English] 11 Standard additional questions for Mathematics Chemistry [English] 11 Standard Maharashtra State Board, and you can use Shaalaa.com to keep it handy for your exam preparation.
