Advertisements
Advertisements
प्रश्न
Identify giving reason whether the following compound is aromatic or not.
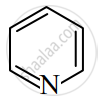
उत्तर
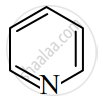
The compound is aromatic since it has 6π electrons and hence, obeys the Huckel rule of aromaticity.
APPEARS IN
संबंधित प्रश्न
Write the balanced chemical reaction to get benzene from Phenol.
Identify the main product of the reaction:
\[\ce{H - C ≡ C - H + H - O ->[{40%} H2SO4][{1%} HgSO4]}\] _____.
Identify A, B, C in the following reaction sequence:
\[\ce{CH3 - CH = CH2 ->[Br2/CCl4][room temperature] A ->[Zn] B ->[dil. alkaline][KMnO4] C}\]
Name two reagents used for acylation of benzene.
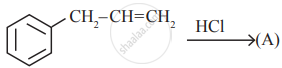 is
is
Which one of the following is non aromatic?
Which of the following can be used as the halide component for friedal - crafts reaction?
2 – butyne on chlorination gives ______.
Describe the mechanism of Nitration of benzene.
Write the chemical equations for combustion of propane.
Suggest the route for the preparation of the following from benzene.
3 – chloro nitrobenzene
Suggest the route for the preparation of the following from benzene.
4 – chlorotoluene
Suggest the route for the preparation of the following from benzene.
Bromo benzene
Suggest the route for the preparation of the following from benzene.
m - dinitro benzene
Write the structure of the following alkanes.
5 – (2 – Ethyl butyl) – 3, 3 – dimethyldecane
Complete the following:
\[\begin{array}{cc}
\ce{CH2 - CH2 ->[Zn/C2H5OH]}\\
|\phantom{.......}|\phantom{..............}\\
\ce{Br}\phantom{.....}\ce{Br}\phantom{..............}
\end{array}\]
How will distinguish 1 – butyne and 2 – butyne?
When acetylene is passed through red hot iron tube at 873 K, ____________ is/are obtained as product/s.
According to Huckel rule, a cyclic π molecular orbital formed by overlap of p orbitals must contain ____________ p electrons.
Which of the following is NOT alicyclic compound?
The alkane formed on heating sodium butanoate with sodalime is ______.
Sodium benzoate on decarboxylation gives ____________.
Read the following reaction and answer the questions given below.
\[\begin{array}{cc}
\phantom{..............................}\ce{CH3}\\
\phantom{...........................}|\\
\ce{CH3 - C = CH2 + HBr ->[benzoyl][peroxide]CH3 - CH - CH2Br}\\
|\phantom{....................................}\\
\ce{CH3}\phantom{.................................}\\
\end{array}\]
- Write the IUPAC name of the product.
- State the rule that governs the formation of this product.
Read the following reaction and answer the questions given below.
\[\begin{array}{cc}
\phantom{...............................}\ce{CH3}\\
\phantom{............................}|\\
\ce{CH3 - C = CH2 + HBr ->[benzoyl][peroxide]CH3 - CH - CH2Br}\\
|\phantom{....................................}\\
\ce{CH3}\phantom{..................................}
\end{array}\]
- Write the IUPAC name of the product.
- State the rule that governs the formation of this product.
Read the following reaction and answer the questions given below.
\[\begin{array}{cc}
\phantom{..........................}\ce{CH3}\\\phantom{........................}|\\\ce{CH3 - C = CH2 + HBr ->[benzoyl][peroxide] CH3 - CH - CH2Br}\\
|\phantom{......................................}\\
\ce{CH3}\phantom{....................................}\end{array}\]
- Write the IUPAC name of the product.
- State the rule that governs the formation of this product.
Read the following reaction and answer the questions given below.
\[\begin{array}{cc}
\phantom{..............................}\ce{CH3}\\
\phantom{............................}|\\
\ce{CH3 - C = CH2 + HBr ->[benzoyl][peroxide] CH3 - CH - CH2Br}\\
|\phantom{....................................}\\
\ce{CH3}\phantom{..................................}
\end{array}\]
- Write IUPAC name of the product.
- State the rule that governs formation of this product.
