Advertisements
Advertisements
प्रश्न
An alkane is obtained by decarboxylation of sodium propionate. Same alkane can be prepared by ______.
विकल्प
Catalytic hydrogenation of propene
action of sodium metal on iodomethane
reduction of 1 – chloro propane
reduction of bromomethane
उत्तर
An alkane is obtained by decarboxylation of sodium propionate. Same alkane can be prepared by action of sodium metal on iodomethane.
APPEARS IN
संबंधित प्रश्न
Predict the possible product of the following reaction.
bromination of phenol
Predict the possible product of the following reaction.
nitration of toluene
Identify the main product of the reaction.
\[\begin{array}{cc}
\ce{CH3 - CH - CH3 ->[{60%}H2SO4][373 K]}\\
|\phantom{............}\\\ce{OH}\phantom{..........}
\end{array}\] _______.
Identify giving reason whether the following compound is aromatic or not.
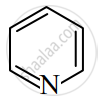
Name two reagents used for acylation of benzene.
Which one of the following is non aromatic?
Identify the compound A, B, C and D in the following series of reactions.

A compound with molecular formula C4H4O has all the four carbon atom and the oxygen atom in the ring. It also has two carbon-carbon double bonds. The compound is ____________.
According to Huckel rule, a cyclic π molecular orbital formed by overlap of p orbitals must contain ____________ p electrons.
Read the following reaction and answer the questions given below.
\[\begin{array}{cc}
\phantom{..............................}\ce{CH3}\\
\phantom{...........................}|\\
\ce{CH3 - C = CH2 + HBr ->[Benzoyl][peroxide] H3C - CH - CH2Br}\\
|\phantom{....................................}\\
\ce{CH3}\phantom{.................................}
\end{array}\]
- Write the IUPAC name of the product.
- State the rule that governs the formation of this product.
