Advertisements
Advertisements
प्रश्न
How will you demonstrate the solubility of ammonia in water? Explain.
उत्तर
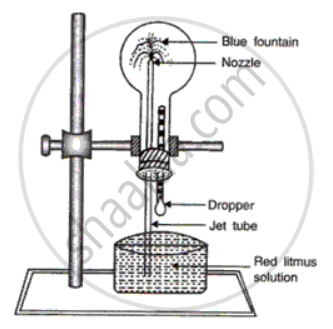
Ammonia is highly soluble in water. This can be demonstrated by Fountain
experiment.
• We take a round bottom flask filled with ammonia gas.
• The flask is closed with a two holed rubber stopper, one for the jet tube and the other for a dropper filled with water.
• The flask is fixed to the stand in an inverted position .
• The free end of the jet tube is dipped into a beaker containing red litmus
solution .
• When the bulb of dropper is pressed, it is observed that red litmus solution rises up and strikes the wall of the flask and spreads in form of fountain, which is blue in colour.
APPEARS IN
संबंधित प्रश्न
Ammonium salts decompose on heating. What other property do ammonium salts have in common?
Give two reactions to show reducing property of ammonia.
Identify the substance underlined, in the following case:
Cation that does not form a precipitate with ammonium hydroxide but forms one with sodium hydroxide.
Identify the substance underlined, in the following case:
A solid formed by reaction of two gaes, one of which is acidic and the other basic in nature.
Given one test can be used to detect the presence of the ion produced
What do you observe when Excess ammonia is mixed with chlorine.
What do you observe when Ammonia in excess is mixed with chlorine.
Choose the correct answer from the options given below :
Ammonia is produced when ammonium chloride is heated with
