Advertisements
Advertisements
प्रश्न
How will you demonstrate the solubility of ammonia in water? Explain.
उत्तर
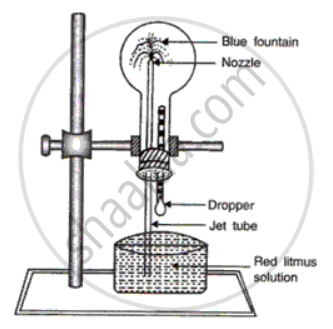
Ammonia is highly soluble in water. This can be demonstrated by Fountain
experiment.
• We take a round bottom flask filled with ammonia gas.
• The flask is closed with a two holed rubber stopper, one for the jet tube and the other for a dropper filled with water.
• The flask is fixed to the stand in an inverted position .
• The free end of the jet tube is dipped into a beaker containing red litmus
solution .
• When the bulb of dropper is pressed, it is observed that red litmus solution rises up and strikes the wall of the flask and spreads in form of fountain, which is blue in colour.
APPEARS IN
संबंधित प्रश्न
Name a metallic chloride soluble in ammonium hydroxide.
Give the formula of (i) Liquid ammonia (ii) Liquor ammonia
List the properties of ammonia that make it
(i) A good refrigerant
(ii) A cleaning agent
(iii) As a source of hydrogen
State your observation for the following cases
(i) Ammonia gas is burnt in an atomosphere of oxygen in the absence of a catalyst
(ii) Glass rod dipped in ammonium hydroxide is brought near the mouth of the concentrated hydrochloric acid bottle
The diagram shows a simple arrangement of the fountain experiment:
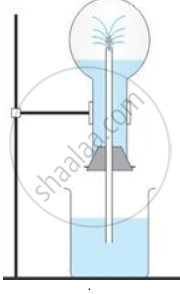
(i) Name the two gases you have studied which can be used in this experiment
(ii) What is the common properly demonstrated by this experiment
Name the other ion formed when ammonia dissolves in water
State one relevant observation of burning of ammonia in air.
What do you observe when Filter paper dipped in colourless phenolphthalein is introduced into ammonia.
What do you observe when Ammonia solution is added drop by drop and then in excess to aqueous copper sulphate solution.
