Advertisements
Advertisements
प्रश्न
Identify condensation polymers and addition polymers from the following.
-(CO(CH2)4 - CONH(CH2)6NH-)n
उत्तर
It is a Condensation polymer.
APPEARS IN
संबंधित प्रश्न
The Zieglar-Natta catalyst is used in the preparation of _______.
(A) LDPE
(B) PHBV
(C) PAN
(D) HDPE
Write the structure of melamine.
Answer the following in one sentence.
Identify 'A' in the following reaction:
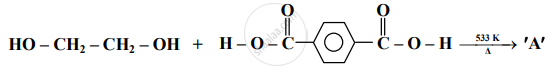
Answer the following in one sentence.
Identify thermoplastic and thermosetting plastic from the following:
- PET
- Urea formaldehyde resin
- Polythene
- Phenol formaldehyde resin
Answer the following.
Write the reaction of the formation of Terylene.
Answer the following.
Name the polymer type in which following linkage is present.
\[\begin{array}{cc}\ce{- C - O -}\\||\phantom{.....}\\
\ce{O\phantom{.....}}\end{array}\]
Draw the structures of polymers formed from the following monomers
\[\ce{n HOOC–R–COOH + n HO–R'–OH}\]
Answer the following.
Draw the structures of polymers formed from the following monomers
H2N–(CH2)5 – COOH
Identify condensation polymers and addition polymers from the following.
-(CH2 - CH = CH - CH2 -)n
Attempt the following:
Write the chemical reactions involved in the manufacture of Nylon 6,6.
Write the reaction involved in the formation of:
Bakelite
Answer the following.
Is synthetic rubber better than natural rubber? If so, in what respect?
Answer the following.
Write main specialities of Buna-S, Neoprene rubber?
Monomer used for preparation of polyacrylonitrile is _____________
write the structure of the monomer used in natural rubber.
Write preparation of low density polythene.
Write chemical reaction for preparation of the following.
Neoprene
Write the name of one example of each polymer in which following repeating units.
\[\begin{array}{cc}
\ce{(-CF2-CF2-), -[NH-(CH2)5-CO] -, -(CH2-CH-), (-CH2-CH2-)}\\
\phantom{............................}|\\
\phantom{..............................}\ce{CN}
\end{array}\]
The following structure represents the polymer:
\[\begin{array}{cc}
\ce{[-C-CH2-NH-C-(-CH2)5 NH -]_{{n}}}\\
\phantom{}||\phantom{.............}||\phantom{................}\\
\phantom{}\ce{O}\phantom{.............}\ce{O}\phantom{................}
\end{array}\]
Identify the CORRECT statement regarding the following polymer.
\[\begin{array}{cc}
\phantom{....}\ce{O}\phantom{............}\ce{O}\phantom{...................}\ce{H}\phantom{.....}\\
\phantom{....}||\phantom{.............}||\phantom{...................}|\phantom{......}\\
\ce{-[C - (CH2)4 - C - NH - (CH2)6 - N -]_{{n}}}
\end{array}\]
Select the CORRECT match for both the polymers.
Novolac is obtained from ____________.
Which of the following polymers is a heteropolymer?
Identify the polymer obtained by polymerization of n moles of acrylonitrile.
Which among the following polymers can NOT be remoulded?
Identify the monomers used in the preparation of Novolac.
Which of the following monomers is used in the manufacture of Neoprene rubber?
Identify addition polymer from the following.
Which of the following polymers is used as insulation for cables?
Which among the following is an example of addition polymer?
The commercial name of polyacrylonitrile is ______.
Match the polymers given in Column I with their repeating units given in Column II.
| Column I | Column II |
| (i) Acrilan |
(a) \[\begin{array}{cc} |
| (ii) Polystyrene | (b) \[\begin{array}{cc} \ce{Cl}\phantom{.......}\\ |\phantom{........}\\ \phantom{}\ce{-(CH2 - C = CH - CH2)\underset{n}{-}} \end{array}\] |
| (iii) Neoprene | (c) \[\begin{array}{cc} \phantom{................................}\ce{CN}\\ \phantom{..............................}|\\ \ce{-(CH2 - CH = CH - CH2 - CH2 - CH)\underset{n}{-}} \end{array}\] |
| (iv) Novolac | (d) \[\begin{array}{cc} \ce{-(CH2 - CH)\underset{n}{-}}\\ \phantom{.....}|\\ \phantom{.......}\ce{CN} \end{array}\] |
| (v) Buna—N | (e) 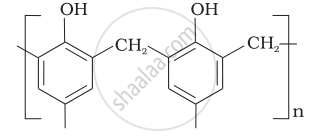 |
| (f) \[\begin{array}{cc} \ce{-(CH2 - CH)\underset{n}{-}}\\ \phantom{.....}|\\ \phantom{......}\ce{Cl} \end{array}\] |
The monomer of Teflon is ______.
Which of the following is an example of polyester?
Which of the following polymers is synthesized using a free radical polymerisation technique?
Nylon threads are made of ______.
Polymer used in bullet proof glass is ______.
Which of the following polymer is used for manufacturing of buckets, dustbins, pipes, etc?
Answer the following.
Name and draw structure of the repeating unit in natural rubber.
Name and draw the structure of the repeating unit in natural rubber.
Another name of terylene is ______.
Name and draw structure of the repeating unit in natural rubber.
Match the following pairs:
| Polymer | Monomer | ||
| (i) | Teflon | (a) | CH2 = CH2 |
| (ii) | PVC | (b) | CF2 = CF2 |
| (iii) | Natural rubber | (c) | Isoprene |
| (iv) | Polythene | (d) | H2C=CHCl |
Name and draw the structure of the repeating unit in natural rubber.
Write the structure of isoprene and the polymer obtained from it.
Name and draw the structure of the repeating unit in natural rubber.
Name and draw structure of the repeating unit in natural rubber.
Write the structure of isoprene and the polymer obtained from it.
