Advertisements
Advertisements
प्रश्न
Identify primary, secondary, tertiary, and quaternary carbon in the following compound.
\[\begin{array}{cc}\ce{CH3}\phantom{..................}\\
|\phantom{....................}\\\ce{CH3 - C - CH - CH2 - CH2 - CH3}\\|\phantom{.....}|\phantom{................}\\
\ce{CH3}\phantom{.}\ce{CH3}\phantom{..............}
\end{array}\]
उत्तर
\[\begin{array}{cc}
\ce{1^\circ}\phantom{...............}\\
\ce{CH3}\phantom{..............}\\
\phantom{.......}|4^\circ\phantom{.}3^\circ\phantom{....}2^\circ\phantom{....}2^\circ\phantom{....}1^\circ\phantom{.}\\
\ce{^{1^\circ}CH3-C-CH-CH2-CH2-CH3}\\
|\phantom{....}|\phantom{............}\\
\ce{CH3\phantom{}CH3}\phantom{.........}\\
\phantom.{}1^\circ\phantom{...}1^\circ\phantom{...........}
\end{array}\]
APPEARS IN
संबंधित प्रश्न
Draw a formula for the first five members of the homologous series beginning with the given compound.
CH3COCH3
Draw a formula for the first five members of the homologous series beginning with the given compound.
H–CH=CH2
Identify the functional group in the following compound.
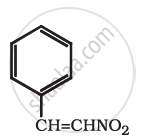
Write the first four members of the homologous series that begins with CH3CHO. Also, write down their general molecular formula.
Structure of the compound whose IUPAC name is 5, 6 – dimethylhept - 2 - ene is
How many cyclic and acyclic isomers are possible for the molecular formula C3H6O?
Describe the classification of organic compounds based on their structure.
Write a note on homologous series.
The reagent used to confirm the presence of five hydroxyl groups in glucose is ____________.
An organic compound containing carbon, hydrogen and oxygen is soluble in dil. H2SO4 and does not react with sodium metal or KMnO4. When it is heated with HI in excess, a single alkyl halide is obtained. The original compound can be ____________.
Which of the following statements is INCORRECT for the organic compounds which belong to homologous series?
Which of the following is NOT a cyclic compound?
How many 3° carbon atoms are present in 1, 4 dibromo-2-methylpentane?
What is the molecular formula of pyran?
How many primary, secondary and tertiary carbon atoms respectively are present in isobutane?
Identify the number of carbon atoms and number of oxygen atoms respectively present in furan molecule.
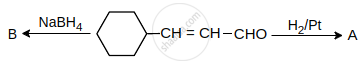
A and B are:
A pyranose ring consists of a skeleton of ______.
Identify primary, secondary, tertiary and quaternary carbon in the following compounds.
\[\begin{array}{cc}
\ce{CH3}\phantom{.................}\\
|\phantom{...................}\\
\ce{CH3 - C - CH - CH2 - CH2 - CH3}\\
\phantom{}|\phantom{....}|\phantom{...............}\\
\phantom{}\ce{CH3}\phantom{.}\ce{CH3}\phantom{.............}
\end{array}\]
Identify primary, secondary, tertiary, and quaternary carbon in the following compounds.
\[\begin{array}{cc}
\ce{CH3}\phantom{..................}\\
|\phantom{....................}\\
\ce{CH3 - C - CH - CH2 - CH2 - CH3}\\
\phantom{}|\phantom{....}|\phantom{................}\\
\phantom{.}\ce{CH3\phantom{.}CH3}\phantom{..............}
\end{array}\]
Identify the α - carbons in the following species and give the total number of
\[\ce{CH3 -CH2 -\overset{⊕}{C}H -CH2 -CH3}\]
Identify primary, secondary, tertiary and quaternary carbon in the following compounds.
\[\begin{array}{cc}
\ce{CH3}\phantom{.................}\\
|\phantom{....................}\\
\ce{CH3 - C - CH - CH2 - CH2 - CH3}\\
|\phantom{....}|\phantom{................}\\
\ce{CH3}\ce{CH3}\phantom{..............}
\end{array}\]
Identify primary, secondary, tertiary, and quaternary carbons in the following compound.
\[\begin{array}{cc}
\ce{CH3\phantom{.................}}\\
|\phantom{....................}\\
\ce{CH3 - C - CH - CH2 - CH2 - CH3}\\
|\phantom{....}|\phantom{................}\\
\ce{CH3 CH3\phantom{.............}}
\end{array}\]
Identify primary, secondary, tertiary and quaternary carbon in the following compound.
\begin{array}{cc}
\ce{CH3}\phantom{..................}\\
|\phantom{....................}\\
\ce{CH3 - C - CH - CH2 - CH2 - CH3}\\
|\phantom{....}|\phantom{................}\\
\ce{CH3}\ce{CH3}\phantom{..............}
\end{array}
Indicate the σ and π bonds in the following molecule:
\[\ce{HCONHCH3}\]
