Advertisements
Advertisements
प्रश्न
If a crop of wheat removes 20 kg of nitrogen per hectare of soil, what mass of the fertilizer, calcium nitrate Ca(NO3)2 would be required to replace the nitrogen in a 10 hectare field?
उत्तर
Total area of the field = 10 hectare
∴ Total nitrogen that has to be replaced = 20 × 10
= 200 kg
Molecular mass of Ca(NO3)2 = 40 + 2 × 62 = 164
164 kg of Ca(NO3)2 contain 28 kg of nitrogen
Now, 28 kg of nitrogen is present in 164 kg of Ca(NO3)2
∴ 200 kg of nitrogen are present in `(164 xx 200)/28` kg of Ca(NO3)2 = 1171.4 kg
APPEARS IN
संबंधित प्रश्न
Give balanced chemical equations for the following conversions A, B, and C:
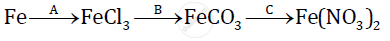
Potassium nitrate on strong heating decomposes as under :
2KNO3 → 2KNO2 + O2
Calculate : Weight of potassium nitrite formed.
(K = 39, 0 = 16, N = 14)
What is the mass of nitrogen in 1000Kg of urea [CO(NH2)2] ?
[H = 1, C= 12, N= 14, O = 16]
Calculate the percentage of boron (B) in borax (Na2B4O7.10H2O). [H = 1, B = 11, O = 16, Na = 23],
answer correct to 1 decimal place.
Washing soda has the formula Na2CO3.10H2O.What is the mass of anhydrous sodium carbonate left when all the water of crystallization is expelled by heating 57.2 g of washing soda?
Define or explain the term:
Vapour density
Calculate the number of atoms of each kind in 5.3 grams of sodium carbonate.
Correct the statement, if required.
Under similar conditions of temperature and pressure, two volumes of hydrogen combined with two volumes of oxygen will give two volumes of water vapour.
Ammonia burns in oxygen and the combustion, in the presence of a catalyst, may be represented by;
\[\ce{2NH3 + 2 1/2O2 -> 2NO + 3H2O}\] [H = 1, N = 14, O = 16]
What mass of steam is produced when 1.5 g of nitrogen monoxide is formed?
The vapour density of CH3OH is ______. (At. Wt. C = 12, H = 1, O = 16)
