Advertisements
Advertisements
प्रश्न
If you take a pinch of sodium hydrogen carbonate powder in a test-tube and add drop-by-drop acetic acid to it, what would you observe immediately? List any two main observations.
उत्तर
When sodium hydrogen carbonate is added to a flask containing acetic acid, the two observations noted are as follows:
(i) Brisk effervescence is seen because of CO2 gas escaping out from the reaction mixture. The gas turns lime water milky.
(ii) Some amount of heat is evolved during the reaction.
Reactions:\[{NaHCO}_3 + {CH}_3 COOH \to {CH}_3 COONa + {CO}_2 \uparrow + H_2 O\]
\[Ca \left( OH \right)_2 + {CO}_2 \to {CaCO}_3 + H_2 O\]
APPEARS IN
संबंधित प्रश्न
Complete the following chemical equations :CH3COOH + Na2CO3 →
Which of the following molecular formula corresponds to ethylbutanoate ester?
(a) C5H10O2
(b) C6H12O2
(c) C7H14O2
(d) C8H16O2
Acetic acid is a typical acid. Write one equation in case of its reactions with a metal?
Name the reducing agent used to convert acetic acid into ethanol?
A student is studying the properties of acetic acid. List two physical properties of acetic acid he observes. What happens when he adds a pinch of sodium hydrogen carbonate to this acid? Write any two observations.
Which of the following observations is true about dilute solution of acetic acid?
(A) It smells like vinegar and turns red litmus blue
(B) It smells like onion and turns blue litmus red
(C) It smells like orange and turns red litmus blue
(D) It smells like vinegar and turns blue litmus red
Give balanced chemical equations for the following conversion:
Calcium carbide to ethyne
Draw the structural diagram of:
Ethanoic acid
Bubbles are seen in the test tube during the preparation of lime water.
Observe the diagram given below and answer the questions:
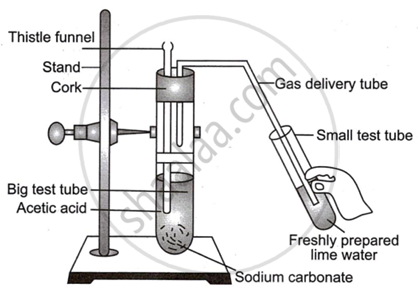
- Name the reactants in this reaction.
- Which gas comes out as effervescence in the bigger test tube?
- What is the colour change in the lime water?
- In the above experiment instead of sodium carbonate which chemical can be used to get same products?
- Write the use of acetic acid.
