Advertisements
Advertisements
प्रश्न
In a cyclotrimetaphosphoric acid molecule, how many single and double bonds are present?
विकल्प
3 double bonds; 9 single bonds.
6 double bonds; 6 single bonds.
3 double bonds; 12 single bonds.
Zero double bonds; 12 single bonds.
उत्तर
3 double bonds; 9 single bonds.
Explanation:
Cyclotrimetaphosphoric acid contains three double bonds and 9 single bonds as shown below: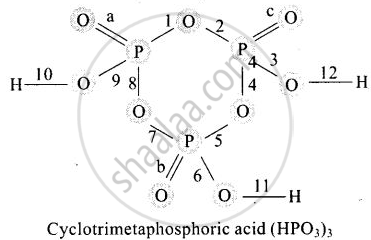
a, b, and c are three pi-bonds and numerics 1 to 12 are sigma bonds.
APPEARS IN
संबंधित प्रश्न
Fe2+ ions react with nitric oxide formed from reduction of nitrate and yields a brown coloured complex _____________.
Calculate molarity and molality of 6.3% solution of nitric acid having density 1.04 g cm−3
. (H = 1, N = 14, O = 16)
What is the action of hot/concentrated nitric acid on - Antimony.
Write the name of gas released when Cu is added to
dilute HNO3
Write the name of gas released when Cu is added to
conc. HNO3
The catalyst used in the manufacture of HNO3 by Ostwald’s process is ____________.
Why is nitric oxide paramagnetic in gaseous state but the solid obtained on cooling it is diamagnetic?
In the ring test of \[\ce{NO^{-}3}\] ion, \[\ce{Fe^{2+}}\] ion reduces nitrate ion to nitric oxide, which combines with \[\ce{Fe^{2+} (aq)}\] ion to form brown complex. Write the reactions involved in the formation of brown ring.
Nitric acid forms an oxide of nitrogen on reaction with P4O10. Write the reaction involved. Also write the resonating structures of the oxide of nitrogen formed.
The number of bond pairs and lone pairs of electrons, on nitrate ions, are respectively:
