Advertisements
Advertisements
Question
In a cyclotrimetaphosphoric acid molecule, how many single and double bonds are present?
Options
3 double bonds; 9 single bonds.
6 double bonds; 6 single bonds.
3 double bonds; 12 single bonds.
Zero double bonds; 12 single bonds.
Solution
3 double bonds; 9 single bonds.
Explanation:
Cyclotrimetaphosphoric acid contains three double bonds and 9 single bonds as shown below: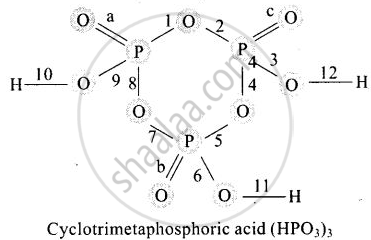
a, b, and c are three pi-bonds and numerics 1 to 12 are sigma bonds.
APPEARS IN
RELATED QUESTIONS
What is the action of hot/concentrated nitric acid on - Arsenic
Illustrate how copper metal can give different products on reaction with HNO3.
Fe2+ ions react with nitric oxide formed from reduction of nitrate and yields a brown coloured complex _____________.
Write the name of gas released when Cu is added to
dilute HNO3
Concentrated nitric acid, upon long standing, turns yellow brown due to the formation of ____________.
Why is nitric oxide paramagnetic in gaseous state but the solid obtained on cooling it is diamagnetic?
\[\ce{P4O6}\] reacts with water according to equation \[\ce{P4O6 + 6H2O -> 4H3PO3}\]. Calculate the volume of 0.1 M NaOH solution required to neutralise the acid formed by dissolving 1.1 g of \[\ce{P4O6}\] in \[\ce{H2O}\].
Nitric acid forms an oxide of nitrogen on reaction with P4O10. Write the reaction involved. Also write the resonating structures of the oxide of nitrogen formed.
Give an example to show the effect of concentration of nitric acid on the formation of oxidation product.
The number of bond pairs and lone pairs of electrons, on nitrate ions, are respectively:
