Advertisements
Advertisements
प्रश्न
In a photoelectric experiment, the stopping potential is 1.5V. What is the maximum kinetic energy of a photoelectron?
उत्तर
Given:
Vs = 1.5 V
To find: Maximum kinetic energy = ?
We know that KEmax = eVs
= 1.6 × 10-19 × 1.5
∴ KE max = 2.4 × 10-19 J
APPEARS IN
संबंधित प्रश्न
Given the following data for incident wavelength and the stopping potential obtained from an experiment on the photoelectric effect, estimate the value of Planck's constant and the work function of the cathode material. What is the threshold frequency and corresponding wavelength? What is the most likely metal used for emitter?
| Incident wavelength (in Å) | 2536 | 3650 |
| Stopping potential (in V) |
1.95 | 0.5 |
Define photoelectric effect.
The energy of a photon is 2 eV. Find its frequency and wavelength.
The maximum velocity of photoelectron emitted is 4.8 m/s. If the e/m ratio of the electron is 1.76 × 1011 C/kg, then stopping potential is given by ______
Which one of the following is TRUE in photoelectric emission?
When a photon enters glass from air, which one of the following quantity does not change?
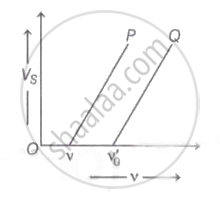
The graph of stopping potential `"V"_"s"` against frequency v of incident radiation is plotted for two different metals P and Q as shown in the graph. ΦP and ΦQ are work-functions of P and Q respectively, then
When light falls on a metal surface, the maximum kinetic energy of the emitted photoelectrons depends upon ______
The maximum velocity of the photoelectron emitted by the metal surface is 'v '. Charge and mass of the photoelectron is denoted by 'e' and 'm' respectively. The stopping potential in volt is ______.
A metal surface is illuminated by light of given intensity and frequency to cause photoemission. If the intensity of illumination is reduced to one-fourth of its original value then the maximum KE of the emitted photoelectrons would be ______.
The work function of a metal is 1.6 x 10-19 J. When the metal surface is illuminated by the light of wavelength 6400 Å, then the maximum kinetic energy of emitted photo-electrons will be (Planck's constant h = 6.4 x 10-34 Js) ____________.
Photoelectrons emitted from a metallic surface are initially ____________.
When wavelength of incident radiation on the metal surface is reduced from 'λ1' to 'λ2', the kinetic energy of emitted photoelectrons is tripled. The work function of the metal is ______.
(h = Planck's constant, c =velocity of light)
Light of different frequencies, whose photons have energies 3 eV and 18 eV respectively, successively illuminate a metal of work function 2 eV. The ratio of the maximum speeds of the emitted electrons will be ______.
The work function of a photosensitive material is 4.0 eV. The longest wavelength of light that can cause photon emission from the substance is (approximately) ____________.
When a photosensitive surface is irradiated by lights of wavelengths `lambda_1` and `lambda_2`, kinetic energies of emitted photoelectrons are E1 and E2 respectively. The work function of the photosensitive surface is ____________.
A light of frequency 'v' is incident on the metal surface whose threshold frequency is 'v0'. If v = v0, then [c = speed of light in medium] ____________.
The radiation corresponding to the 3 → 2 transition of a hydrogen atom falls on a gold surface to generate photoelectrons. These electrons are passed through a magnetic field of 5 × 10-4 T. Assume that the radius of the largest circular path followed by these electrons is 7 mm, and the work function of the metal is ______.
(Mass of electron = 9.1 × 10-31 kg)
When radiation of wavelength λ is used to illuminate a metallic surface, the stopping potential is V. When the same surface is illuminated with radiation of wavelength 3λ, the stopping potential is `"V"/4`. If the threshold wavelength for the metallic surface is nλ. then value of n will be ______.
Photoelectrons are observed to just emit out of a material surface when the light of 620 nm falls on it with the intensity of 100 W m-2. If the light of wavelength 400 nm is incident on the same material with an intensity of 1 W m-2, what would be the minimum reverse potential needed to stop the outflow of the electrons?
The wavelength of light incident on a metal surface is reduced from 300 nm to 200 nm (both are less than threshold wavelength). What is the change in the stopping potential for photoelectrons emitted from the surface will be ______ V. (Take h = 6.6 × 10-34 J-s)
When ultraviolet light of wavelength 100 nm is incident upon a sample of silver metal, a potential difference of 7.7 volt is required to stop the photoelectrons from reaching the collector plate. The potential required to stop photo electrons when light of wavelength 200 nm is incident upon silver is ______.
The maximum kinetic energy of the photoelectrons ejected will be ______ eV when the light of wavelength 350 nm is incident on a cesium surface. The work function of cesium = 1.9 eV.
A charged dust particle of radius 5 × 10-7 m is located in a horizontal electric field having an intensity of 6.28 × 105 V/m. The surrounding medium is air with a coefficient of viscosity η = 1.6 × 10-5 N-s/m2. If the particle moves with a uniform horizontal speed of 0.02 m/s, the number of electrons on it is ______.
On a photosensitive material when frequency of incident radiation is increased by 30%, kinetic energy of emitted photoelectrons increases from 0.4 eV. The work function of the surface is ______.
The photoelectric threshold for a certain metal surface is 3600 Å. If the metal surface is irradiated by a wavelength of 1100 Å, then kinetic energy of the emitted photoelectrons is ______.
When monochromatic light of frequency v1 falls on a metal surface, the stopping potential required is found to be V1. If the radiation of frequency v2 is incident on the surface, the stopping potential required V2 is ______. (v2 > v1)
If the maximum kinetic energy of emitted electrons in the photoelectric effect is 2eV, the stopping potential will be ______.
Give Einstein's explanation of the photoelectric effect.
