Advertisements
Advertisements
प्रश्न
In the following structure, the percentage of the 's' character in the lone pair occupy by the oxygen atom is ______.
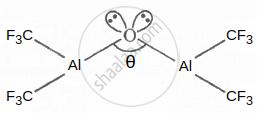
Given: Cos θ = −0.99
विकल्प
0.26
1.89
2.64
3.12
MCQ
रिक्त स्थान भरें
उत्तर
In the following structure, the percentage of the 's' character in the lone pair occupy by the oxygen atom is 0.26.

Explanation:
Cos θ = `"s"/("s" - 1)` = −0.99
s = `0.99/1.99 xx 100` = = 49.74%
% of the 's' character in lone pair by oxygen = 2%
s = 100 – (2 × 49.74)
⇒ % of the s = 0.26
shaalaa.com
क्या इस प्रश्न या उत्तर में कोई त्रुटि है?
