Advertisements
Advertisements
Question
In the following structure, the percentage of the 's' character in the lone pair occupy by the oxygen atom is ______.
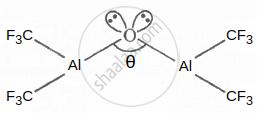
Given: Cos θ = −0.99
Options
0.26
1.89
2.64
3.12
MCQ
Fill in the Blanks
Solution
In the following structure, the percentage of the 's' character in the lone pair occupy by the oxygen atom is 0.26.

Explanation:
Cos θ = `"s"/("s" - 1)` = −0.99
s = `0.99/1.99 xx 100` = = 49.74%
% of the 's' character in lone pair by oxygen = 2%
s = 100 – (2 × 49.74)
⇒ % of the s = 0.26
shaalaa.com
Is there an error in this question or solution?
