Advertisements
Advertisements
प्रश्न
In which of the following pairs, the two species are isostructural:
विकल्प
\[\ce{SO^{2-}_3}\] and \[\ce{NO^-_3}\]
BF3 and NF3
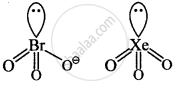
\[\ce{BrO^-_3}\] and \[\ce{XeO3}\]
SF4 and XeF4
MCQ
उत्तर
\[\ce{BrO^-_3}\] and \[\ce{XeO3}\]
Explanation:
Both are pyramidal.
shaalaa.com
क्या इस प्रश्न या उत्तर में कोई त्रुटि है?
