Advertisements
Advertisements
प्रश्न
It has been observed that first ionization energy of 5 d series of transition elements are higher than that of 3d and 4d series, explain why?
उत्तर
The transition elements of 5d series have intervening 4f orbitals. There is greater effective nuclear charge acting on outer valence electrons due to the weak shielding by 4f electrons. Hence first ionisation energy of 5d series of transition elements are higher than that of 3d and 4d series.
APPEARS IN
संबंधित प्रश्न
How would you account for the following : Transition metals form coloured compounds
For M2+/M and M3+/M2+ systems, the EΘ values for some metals are as follows:
| Cr2+/Cr | −0.9 V |
| Mn2+/Mn | −1.2 V |
| Fe2+/Fe | −0.4 V |
| Cr3/Cr2+ | −0.4 V |
| Mn3+/Mn2+ | +1.5 V |
| Fe3+/Fe2+ | +0.8 V |
Use this data to comment upon:
The stability of Fe3+ in acid solution as compared to that of Cr3+ or Mn3+.
The transition metals show _________ character because of the presence of unpaired· electrons and Cu+ is ____________ because of its electronic configuration is [Ar]3d10
When \[\ce{KMnO4}\] solution is added to oxalic acid solution, the decolourisation is slow in the beginning but becomes instantaneous after some time because ______.
Identify the metal and justify your answer.
\[\ce{MO3F}\]
A complex in which dsp2 hybridisation takes place is
How is the variability in oxidation states of transition metals different from that of p-block elements?
Which property of transition metals enables them to behave as catalysts?
The trend of which property is represented by the following graph?
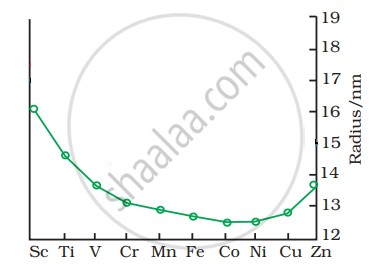
Compare the general characteristics of the first series of the transition metals with those of the second and third series metals in the respective vertical columns. Give special emphasis on the following point:
Atomic sizes
