Advertisements
Advertisements
प्रश्न
List the conclusions you will draw while studying the following properties of ethanoic acid :
(a) Odour
(b) Solubility in water
(c) Effect on litmus paper
(d) Reaction with sodium hydrogen carbonate
उत्तर
Properties of Ethanoic Acid (CH3COOH)
(a) Odour : It has pungent smell.
(b) Solubity in water It is soluble in water due to the formation of hydrogen bonding.
(c) Effect on litmus paper
∗ It turns blue litmus red, indicating that it is acidic in nature
CH3COOH \[\rightleftharpoons\] `CH_3COO^(-) + H^+`
∗ When dissolved in water, acetic acid undergoes dissociation to form `H^+` ion . Due to the release of `H^+` , CH3COOH is acidic in nature .
(d) Reaction with sodium hydrogen carbonate
CH3COOH + NaOHCO3 → CH3COONa + CO2(g) + H2O(l)
APPEARS IN
संबंधित प्रश्न
Which of the following molecule is called buckministerfullerene?
C90 C60 C70 C120
Choose the correct alternative
The process by which decayed plants slowly convert into coal is called.
TRUE \ FALSE
Organic compounds are soluble in water.
Name of the following compounds :
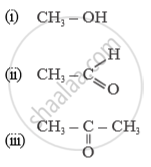
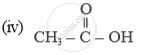
Answer the following question.
Name the type of bonds formed in the methane compound.
Answer the following question.
Why are methane compounds have low melting and boiling points?
Starting from carbon dioxide how would you obtain a fertilizer.
[Give balanced equations for the same]
The portion left on dropping a hydrogen atom from an alkane is called ______.
Carbon is one of the most important ______ elements.
According to the Octet rule, carbon requires ______ electrons to attain the nearest noble gas.
